Summary
- Potassium iodide may be used during radiological or nuclear emergencies to help protect the thyroid from absorbing radioactive iodine.
- Potassium iodide tablets should only be taken when advised by public authorities. Medical advice is recommended for those with thyroid-related conditions.
- Only take the recommended dose for your age group (see dosage table)
- Potassium iodide only protects the thyroid from exposure to radioactive iodine and does not protect other parts of your body or against other forms of radiation.
- An example of an incident that may involve the release of radioactive iodine is an emergency at a nuclear reactor.
Note to readers
This advisory note provides clear guidance on the use of potassium iodide tablets during a radiological or nuclear emergency as referred to in Radiation Protection Series (RPS) G-3: Guide for Radiation Protection in Emergency Exposure Situations.
The target audience of this advice is the Australian public, and Australians residing or travelling overseas.
Radiological or nuclear emergencies and actions
Radiological or nuclear emergencies and actions
Radiological or nuclear emergency in Australia
In the rare event of a radiological or nuclear emergency in Australia, the decision to distribute potassium iodide tablets will be made by state or territory authorities.
The relevant public authorities for potassium iodide distribution should be identified in state and territory emergency response plans and arrangements.
Radiological or nuclear emergency overseas
If a radiological or nuclear emergency occurs overseas, Australian authorities will work closely with international partners to assess the situation and provide guidance.
ARPANSA will advise the Department of Foreign Affairs and Trade (DFAT) on whether potassium iodide tablets should be used by Australians overseas. DFAT are the Australian government coordinating agency for international crises as designated within the Australia Government Crisis Management Framework.
Unless you receive advice from Australian authorities, you should always follow the instructions of local emergency services and public health authorities in the country you are in or travelling through. They will have the most up-to-date information and are best placed to guide you on protective actions.
It is recommended to stay informed through the Smartraveller website. When an emergency occurs, DFAT will also set up an online crisis registration portal (via the DFAT website). This will allow you to receive emergency updates and register your location and contact details.
What is a radiological or nuclear emergency?
A radiological or nuclear emergency is an emergency in which there is a risk of radiation exposure due to the release or potential release of radioactivity.
What is an urgent protective action?
A protective action is an action that can be taken for the purposes of avoiding or reducing radiation exposure in an emergency.
In the event of a radiological or nuclear emergency, an urgent protective action must be taken promptly (usually within 24 hours) to be effective. Figure 1 shows the types of urgent protective actions that could be taken following such an incident.
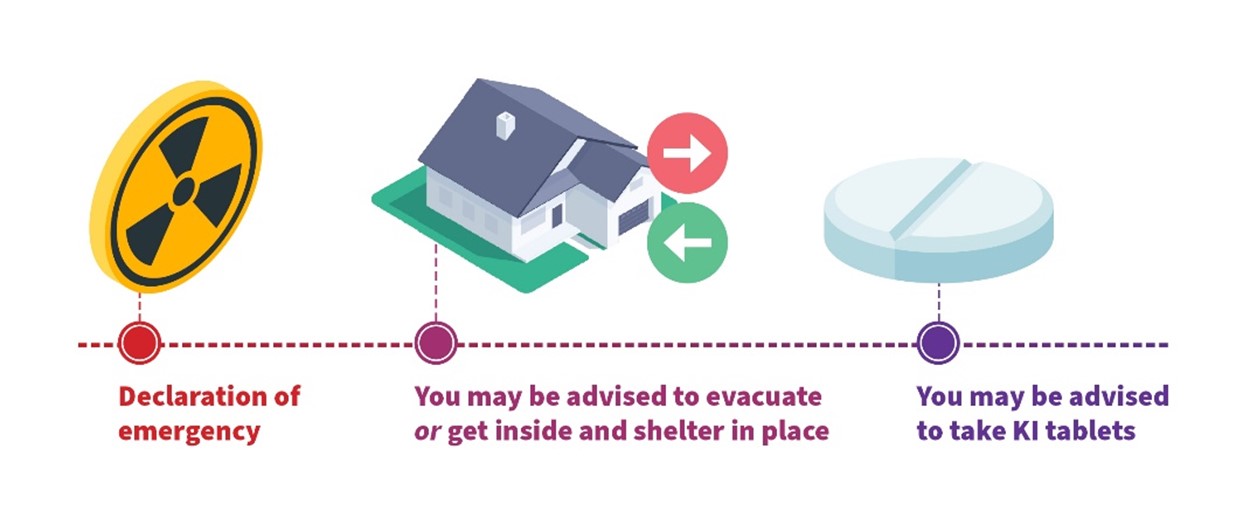 Figure 1: Examples of urgent protective actions that could be taken following the declaration of an emergency.
Figure 1: Examples of urgent protective actions that could be taken following the declaration of an emergency.
Urgent protective action: Iodine thyroid blocking
Iodine thyroid blocking using potassium iodide (KI) is an example of a short-term, urgent protective action. It may be implemented as a precaution, as a standalone action or combined with other protective actions such as sheltering. Only take potassium iodide when advised by public authorities.
In general, potassium iodide should be taken:
- if exposure due to radioactive iodine is expected
- before or shortly after a release of radioactive iodine
- only within a short period before or after the intake of radioactive iodine
- in accordance with the advice of the public authority
- with consideration of individual health concerns (such as pregnancy, breastfeeding and medical conditions).
Potassium iodide advice
What are potassium iodide tablets?
Potassium iodide (KI) tablets contain stable iodine, a type of iodine that is not radioactive. They are used in radiological or nuclear emergencies involving releases of radioactive iodine. If used appropriately, potassium iodide can help block radioactive iodine from being absorbed by the thyroid.
Potassium iodide is available in an easy to swallow tablet form and can be crushed into an oral solution for newborns (0 to 28 days), infants (1 month to 1 year), toddlers (1 to 3 years) and children (3 to 12 years).
What are the potential health effects from exposure to radioactive iodine during a radiological or nuclear emergency?
If people breathe in radioactive iodine, or swallow food or water contaminated with radioiodine, their thyroid gland can absorb the radioactive iodine just like regular iodine. The primary health effect of concern from exposure of the thyroid to radiation is an increase in the risk of developing thyroid cancer in future, especially for children and young adults who are more sensitive to its effects. It could also potentially increase the risk for thyroid problems including hypothyroidism (underactive thyroid) or hyperthyroidism (overactive thyroid).
In addition, if livestock such as dairy animals are exposed and consume grass contaminated with radioactive iodine, the radioactive iodine will be passed into their milk. People could be exposed to radioactive iodine from drinking the milk or eating dairy products made from contaminated milk.
Why do we use potassium iodide tablets?
The thyroid gland plays a vital role in growth and development and regulating the body’s metabolism. It does this by absorbing iodine from the bloodstream to produce essential hormones. In a radiological or nuclear emergency, radioactive iodine may be released into the air. If used appropriately, potassium iodide can help block radioactive iodine from being absorbed by the thyroid.
It is likely that only people near the site of a radiological or nuclear emergency are going to need potassium iodide. Public authorities will advise them if it is necessary to take potassium iodide. Those located further away are unlikely to be exposed to harmful levels of radioactive iodine and should not take potassium iodide unless advised by authorities.
When taken at the correct time, potassium iodide tablets reduce the uptake of radioactive iodine in the thyroid, reducing the risk of thyroid cancer.
How do potassium iodide tablets work?
When a person takes a potassium iodide tablet, it fills the thyroid with stable iodine that prevents the thyroid gland from absorbing any of the radioactive iodine released into the environment. If used appropriately, the thyroid becomes full of stable iodine and is unable to absorb any more iodine for the following 24 hours. See Figure 2.
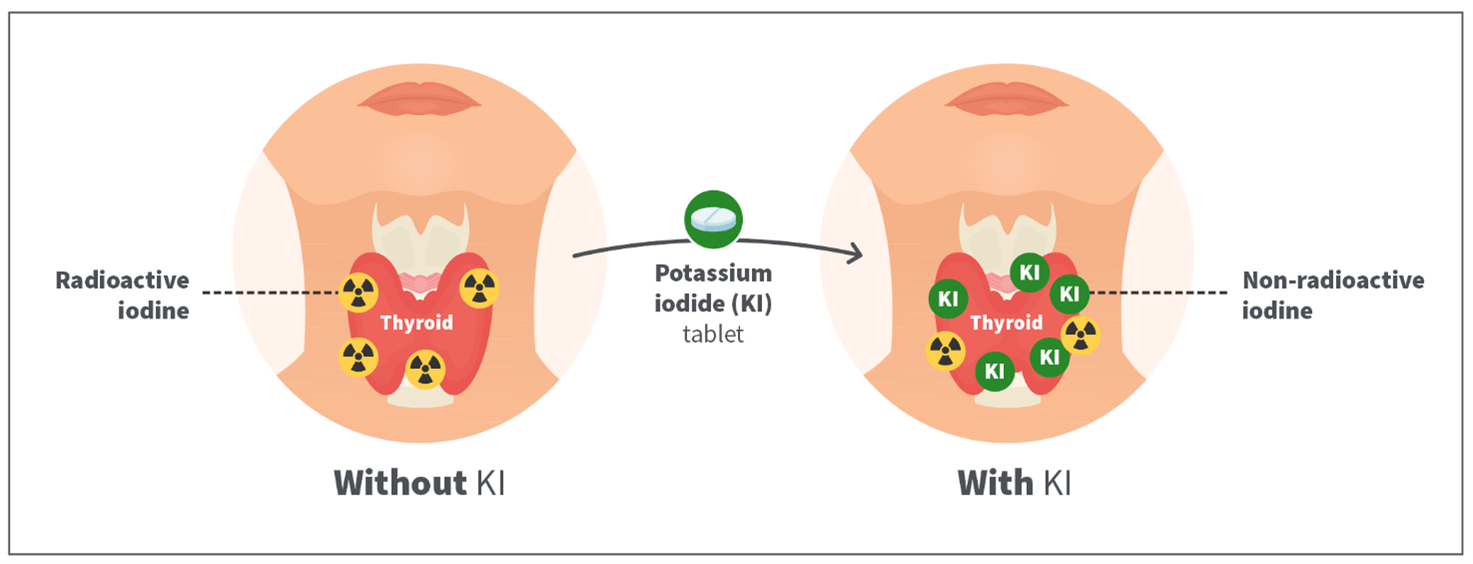 Figure 2: How potassium iodide tablets work when consumed.
Figure 2: How potassium iodide tablets work when consumed.
Children, adolescents and pregnant women are most likely to benefit from potassium iodide as developing thyroids have an increased risk of thyroid cancer from radioactive iodine exposure. Individuals over 40 years of age are likely to receive less benefit from potassium iodide.
If the thyroid gland is damaged, potassium iodide tablets cannot reverse the adverse effects caused by radioactive iodine.
Does potassium iodide protect me from all radiation?
No. Potassium iodide will only protect your thyroid from radioactive iodine and does not protect other parts of your body. It only protects the thyroid if there is a risk of internal exposure. In other words, ingestion or inhalation to radioactive iodine. It will not protect you from other radioactive substances or external exposure of your body to radiation.
Potassium iodide tablets are one form of an urgent protective action and should be performed with other protective actions such as sheltering, stopping consumption of contaminated food and water, and evacuation or relocation on the advice of public authorities.
When and how much potassium iodide should I take?
You should only take potassium iodide tablets if advised by the public authority.
If you have certain medical conditions, including a family history of thyroid conditions, kidney disease or renal dysfunction or possible iodine sensitivity, please make this information known to the public authorities before potassium iodide consumption, as you may need specific medical advice.
 Figure 3: Potassium iodide dosages per age group.
Figure 3: Potassium iodide dosages per age group.
Potassium iodide may be available in different forms and strengths, including:
- Tablets (commonly 65 mg or 130 mg)
- Commercial oral (paediatric) solution, which is a ready-made liquid.
If a commercial oral solution is not available or if tablets cannot be swallowed, a homemade oral solution can be prepared from tablets. Instructions for preparing a homemade solution are provided in Appendix A: Preparation of potassium iodide solution from tablets for infants, children and young adults.
Potassium iodide tablets or liquids bought from a chemist or pharmacy may not be the same strength as the recommended dose. Products can come in different tablet sizes, strengths or liquid concentrations. Always check the label carefully and follow the instructions provided with the product.
The recommended intake amount per age group for tablets and oral solutions when advised by the public authority is shown below.
Table 1: Recommended intake amount per age group
Age group | Potassium iodide dose (mg) | Number or fraction of 130 mg tablets | Number or fraction of 65 mg tablets | Liquid amount (based on 65 mg per mL solution**) |
| Newborns (birth to 1 month) | 16 | 1/8* | ¼* | 0.25 mL |
| Infants or Toddlers (1 month to 3 years) | 32 | ¼* | ½* | 0.5 mL |
| Children (3 to 12 years) | 65 | ½ | 1 | 1 mL |
| Adolescents (12 to 18 years) | 130 | 1 | 2 | 2 mL |
| Adults (over 18 years) | 130 | 1 | 2 | 2 mL |
| Pregnant and breastfeeding women | 130 | 1 | 2 | 2 mL |
*If tablets cannot be swallowed, a homemade oral solution can be prepared from tablets. Instructions for preparing a homemade solution are provided in Appendix A: Preparation of potassium iodide solution from tablets for infants, children and young adults.
**Liquid amounts only apply when using a commercial oral solution that is 65 mg per mL. Always check the label before administering. Homemade oral solutions are provided in the instructions in Appendix A if you'd prefer to consume protective potassium iodine in liquid form instead of tablet.
If you are unsure of your recommended dosage, how to prepare a solution or give it, please contact the relevant public authority.
When does potassium iodide need to be administered?
The optimal period of administration of potassium iodide is:
- less than 24 hours prior to the expected onset of radioactive iodine exposure
- up to 4 hours after radioactive iodine exposure.
The use of potassium iodide may still provide some protection 8 hours after the exposure. Between 8 to 24 hours after exposure, potassium iodide may only provide very limited protection. Commencing potassium iodide later than 24 hours after the exposure is not recommended.
How long does the protective effect of potassium iodide last?
Taking potassium iodide once is usually sufficient to protect your thyroid gland for about 24 hours. However, repeated administration of potassium iodide may be necessary in the case of prolonged (beyond 24 hours) or repeated exposure, unavoidable ingestion of contaminated food and drinking water, and where evacuation is not feasible.
Repeated administration may pose additional risks to newborns (0-1 month), pregnant and breastfeeding women and people older than 60 years and should only be undertaken if advised. Newborns are particularly at risk for intellectual impairment secondary to induced hypothyroidism (cretinism) if repeated doses are administered and should only do so under medical supervision with follow-up thyroid function tests.
When can I stop using potassium iodide tablets?
You will be advised by the public authority to stop taking potassium iodide tablets.
The administration of potassium iodide should cease when the outdoor air no longer contains significant amounts of radioactive iodine. If you experience the following symptoms, discontinue use and seek immediate medical attention:
- burning sensation in mouth or throat
- sore teeth and gums
- rash or hives
- difficulty breathing or swollen salivary glands
- inflammation of the thyroid
- swelling of the face, lips or tongue
- fever
- sore muscles and joints.
What follow-up monitoring or treatment is required after taking potassium iodide tablets?
After the radiological or nuclear emergency, the relevant public authority may advise on a medical follow-up due to radiation exposure.
If you have a known history of thyroid conditions, kidney disease or renal dysfunction and you notice a change in your condition, you should seek immediate medical advice.
If newborns have taken potassium iodide or if you are pregnant, you should inform your doctor to add the dose of potassium iodide to your medical records and assess the thyroid function of your baby.
Other individuals will not need specific follow-up monitoring or treatment. However, if medical attention is needed for other reasons, you should advise a medical professional that you have taken potassium iodide tablets.
What if I am pregnant?
You should only take potassium iodide tablets when instructed by the relevant public authority and follow the recommended dosage for adults.
If instructed, you should only take potassium iodide once. When the radiological or nuclear emergency is over, you should inform your doctor to add the dose of potassium iodide to your medical records and assess the thyroid function of your newborn baby.
What if I am breastfeeding?
You should only take potassium iodide when instructed by the relevant public authority and follow the recommended dosage for adults.
When you are advised to take potassium iodide, some of the iodine passes into breast milk to protect your breastfed child. However, potassium iodide tablets do not ensure protection of breastfed children from other radionuclides that may transfer to breast milk.
If you have been exposed to radiological contamination, you may need to consider minimising breastfeeding for a period from several hours to several weeks, depending on the type and amount of radiation you have been exposed to. In many cases, expressing and discarding breast milk during this period is advised to maintain milk supply. Since reducing or stopping breastfeeding can lead to a decline in milk production and may result in a transition to formula feeding, it is important to consult your midwife, general practitioner or lactation consultant for personalised guidance and support. If practical, it may be advisable to use previously expressed milk stored before the emergency, infant formula (with bottled water if possible), or available substitutes for a period of time.
If you do not have stored breast milk or formula, continue breastfeeding. Wash your hands, nipples, and breasts thoroughly with soap and warm water before breastfeeding or expressing milk.
If you or your baby has a thyroid condition, it is recommended you seek medical advice.
Are there side effects from taking potassium iodide?
When used as instructed by the relevant public authority, the overall benefits of thyroid blocking with potassium iodide during a nuclear emergency will outweigh the risks of side effects. When the appropriate dosage is followed, side effects from potassium iodide are rare. However, they may include mild gastrointestinal upset (for example, nausea or diarrhoea) and skin rash. Never take more than instructed. Larger doses will not provide more protection and may increase the risk of side effects. Potassium iodide should not be taken on a precautionary ‘just in case’ basis or for psychological reassurance.
Caution should be exercised to those who have thyroid conditions (examples include multinodular goitre, Grave’s Disease, or autoimmune thyroiditis), kidney disease or renal dysfunction. If you have these conditions, please seek medical advice.
Iodine is a nutrient our body needs and is found in a wide range of foods such as seafood, eggs and dairy. The amount of potassium iodide used in a radiological or nuclear emergency is much higher than what we normally eat in a day and may cause some symptoms of toxicity with longer term use. Iodine toxicity is uncommon, especially when potassium iodide is used for only a short time as instructed in an emergency.
It is important to note that you cannot be allergic or sensitive to iodine itself. However, if you have uncertainty about possible sensitivity to iodine from reactions to iodine-containing products in the past, you may need specific medical advice before taking potassium iodide.
Can I consume table salt or use dietary supplements containing iodine as a substitute for potassium iodide?
No. Do not use table salt, dietary supplements or foods that contain iodine as a substitute for potassium iodide. These foods do not have enough stable iodine to block radioactive iodine from getting into your thyroid gland. Eating table salt and dietary supplements in large amounts could be harmful.
Another compound that authorities may decide to use as a viable alternative is potassium iodate. This will be given under the instruction of authorities with the appropriate dosage being indicated.
Appendix A: Preparation of potassium iodide solution from tablets for infants, children and young adults
Guidance for home preparation of potassium iodide solution using 130 mg tablet
You will need:
- 1 x 130 mg tablet
- teaspoon
- small bowl
- water
- drink solution of choice: milk, juice, soft drink, infant formula or cordial.
Crush the tablet.
Put one 130 mg potassium iodide tablet in a small bowl and grind into a fine powder with the back of the teaspoon. There should be no large pieces.
Soften the ground tablet powder.
- Add 4 teaspoons (20 mL) of water to the potassium iodide powdered tablet. Mix them together until the potassium iodide powder is dissolved in the water.
- Select a drink from the list above. Prepare 4 teaspoons amount of the drink that can be fully consumed (i.e. small cup of milk, juice or water for a child).
- Add the potassium iodide and water mix to the selected drink.
- Stir to fully mix the potassium iodine, water and drink mixture and assist the infant, child or young adult to consume the full quantity of the drink. It is now the final potassium iodide solution.
Consume the right amount of the final potassium iodide solution using the table below:
Age | Recommended dose of potassium iodide solution |
Newborn to 1 month | 1 teaspoon |
1 month to 3 years | 2 teaspoons |
3 to 12 years | 4 teaspoons |
The mixture can be kept up to 1 week, if refrigerated. After one week, discard the unused portion and prepare a fresh mixture.
Guidance for home preparation of potassium iodide solution using 65 mg tablet
You will need:
- 1 x 65 mg tablet
- teaspoon
- small bowl
- water
- drink solution of choice: milk, juice, soft drink, infant formula or cordial.
Crush the tablet.
- Put one 65 mg potassium iodide tablet in a small bowl and grind into a fine powder with the back of the teaspoon. There should be no large pieces.
Soften the ground tablet powder.
- Add 4 teaspoons (20 mL) of water to the potassium iodide powdered tablet. Mix them together until the potassium iodide powder is dissolved in the water.
- Select a drink from the list above. Prepare 4 teaspoons of the drink that can be fully consumed (i.e. small cup of milk, juice or water for a child).
- Add the potassium iodide and water mix to the selected drink.
- Stir to fully mix the potassium iodine, water and drink mixture and assist the infant, child or young adult to consume the full quantity of the drink. It is now the final potassium iodide solution.
Consume the right amount of the final potassium iodide solution using the table below:
Age | Recommended dose of potassium iodide solution |
Newborn to 1 month | 2 teaspoons |
1 month to 3 years | 4 teaspoons |
3 to 12 years | 8 teaspoons |
The mixture can be kept up to 1 week, if refrigerated. After one week, discard the unused portion and prepare a fresh mixture.
Additional resources
- ARPANSA (2019) RPS G-3 Guide for Radiation Protection in Emergency Exposure Situations, ARPANSA website, accessed 03 November 2025.
- Department of Health, Disability and Ageing (2012), Australian Clinical Guidelines for Radiological Emergencies, Department of Health, Disability and Ageing website, accessed 03 November 2025.
- Department of the Prime Minister and Cabinet (2025), Australian Government Crisis Management Framework (AGCMF), PM&C website, accessed 03 November 2025.
- World Health Organization (2017), Iodine thyroid blocking: Guidelines for use in planning and responding to radiological and nuclear emergencies, WHO website, accessed 03 November 2025.


