What is the Australian Clinical Dosimetry Service (ACDS)?
The ACDS is a world-class dosimetry auditing program assisting in the safe treatment of cancer via radiation therapy in Australia.
Our audits ensure radiation doses are delivered with accuracy during radiation therapy.
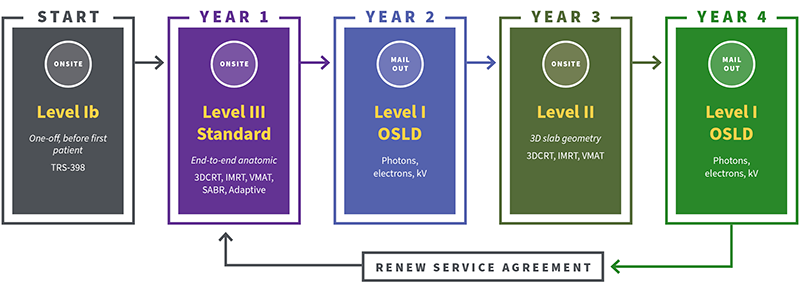
We offer a four-year auditing subscription to radiotherapy providers with differing levels of audits available.
Information gathered during ACDS audits provides data for benchmarking between radiation therapy facilities. We offer radiotherapy audit results across Australia and New Zealand. The program has operated since February 2011.
Our vision
We strive to be a world-leading dosimetry auditing service. Providing the highest level of quality assurance and patient safety in radiation therapy.
Our mission
We aim to guide, support, and improve patient safety and radiotherapy service delivery by:
- providing a comprehensive suite of audit modalities covering all common clinical practices
- improving national dosimetry capabilities in clinical treatment delivery
- offering our services to Australian and overseas radiotherapy centres on a fee-for-service basis.
Our mission aligns with Key Activity 1 of ARPANSA. To ‘Initiate, maintain, and promote frameworks for protection and safety’.
How does the service work?
If you are a radiation therapy clinic treating cancer, you're required to comply with different audits across 4 years. To start auditing your practice:
1. email acds@arpansa.gov.au or call +61 3 9433 2220 with your clinic's details and address
2. pay the fee by following the instructions we send you
3. wait for your first audit instructions, your testing device will be sent via mail.
Learn more about Level Ib audits
The onsite Level Ib audit offers a more accurate measurement of the linac output than the Level I audit and determines the absorbed dose to water per monitor unit for megavoltage photon and electron beams under reference conditions. We visit your facility and perform onsite measurements in your own water tank. All other dosimetry equipment is brought in by us, ensuring an independent audit
We are ISO/IEC 17025 accredited by the National Association of Testing Authorities (NATA) which offers dosimetry audits to meet:
- Radiation Oncology Practice Standards (ROPS)
- Radiation Oncology Health Program Grants (ROHPG) funding
- Trans-Tasman Radiation Oncology Group (TROG) clinical trial credentialing
- Jurisdictional radiation license requirements.
Radiation Oncology Practice Standards
Our audits enables dosimetric inter-comparison. As outlined in the Radiation Oncology Practice Standards (ROPS) Part A, Standard 15, Appendix 5; Part B, Standard 15.
The audit program ensures compliance with the ROPS:
- ensuring radiation doses are accurately delivered in a radiation therapy facility
- aligned with the requirements of ROHPG.
Trans-Tasman Radiation Oncology Group (TROG) & Clinical Trial Credentialling
Our audits may be used towards clinical trial accreditation for TROG trials. Requirements for each individual trial may vary, usually at the discretion of the trial physicist.
Please contact qa@trog.com.au for further information.
Affiliations:
Global Harmonization Group (GHG)
We are an observer member of the GHG, a group that ensures quality in radiation therapy clinical trials. The group consists of:
- clinical trial quality assurance offices
- auditing bodies around the world.
The GHG group aims to harmonise and improve clinical trial QA in radiation therapy worldwide.
Imaging and Radiation Oncology Core (IROC) Houston
We have a Memorandum of Understanding of Mutual Recognition of dosimetric audit equivalence for the Level I OSLD audits. The agreement is with the and IROC Houston Quality Assurance Center. It recognises:
- technical equivalence
- the frequency of both organisations’ OSLD mail out audit.
This means our Level I OSLD results may be used for an IROC OSLD audit, such as a clinical trial. This agreement is maintained by regular inter-comparisons of the mail out audits between us and IROC.
Contact us
For all enquiries, please get in touch via email or phone:
Email: acds@arpansa.gov.au
Phone: +61 3 9433 2220
See below for more information about the ACDS