Part 1: CEO's Review – Dr Carl-Magnus Larsson
This Page:
- International best practice - relevance to Australia and ARPANSA

- Major licence applications from ANSTO
- Status of radiation safety among licence holders
- Incident reporting in Australia
- Regulatory governance
- Financial performance
- Outlook
I am pleased to present to you the sixteenth Annual Report of the CEO of ARPANSA, the fifth under my stewardship of the Agency.
I have the pleasure of working with people who have a strong commitment to the protection of the Australian people and environment from the harmful effects of radiation. Over this year we have, in accordance with statutory obligations:
- provided advice and services to the Australian community on radiation protection, nuclear safety, security, and medical exposures to radiation, including related research
- promoted national uniformity of radiation protection and nuclear safety policy and practices across the Australian Government and states and territories, and
- independently regulated the radiation sources, radiation facilities and nuclear installations of Australian Government entities and contractors.
I am pleased to report that we met the majority of our specific commitments to government. Details on what has been achieved can be found in Part 3: Report on Performance. We did not meet the target set for the length of time taken to assess facility and source licence applications due to the complexity of new applications for nuclear installations and prescribed radiation facilities received from the Australian Nuclear Science and Technology Organisation (ANSTO) simultaneously.
In this review, I take the opportunity to present some significant developments in radiation protection and nuclear safety during the year, as well as some challenges for the Agency as we enter the next financial year.
International best practice – relevance to Australia and ARPANSA
ARPANSA’s engagement with the international radiation safety community is substantial and also vital to the Agency’s capability to implement international best practice in radiation protection and nuclear safety/security in Australia. I review some aspects of this engagement and its domestic relevance below.
Standards, Guidance and Recommendations
ARPANSA participates in the work of the International Atomic Energy Agency (IAEA) Safety Standards Committees as well as in the IAEA Nuclear Security Guidance Committee. Of these Committees, ARPANSA (through Dr Geoff Williams) chairs the Waste Safety Standards Committee; I am a member of the Commission on Safety Standards which oversees the general direction of the Standards development.
The work in the Committees and the Commission on Safety Standards is of direct relevance to the implementation of international best practice across all Australian jurisdictions. One of my statutory obligations is to promote national uniformity in radiation protection and nuclear safety, and for this purpose the Radiation Health Committee has been established with regulators from all Australian jurisdictions. The Radiation Health Committee has agreed to adopt, to the extent practicable, the Safety Standards and Security Guidance developed by the IAEA. To this end, ARPANSA has improved its procedures for contributing to, and commenting on, draft standards and guidance, in consultation with state and territory regulators and other stakeholders. This will improve the effectiveness and efficiency of implementation of international best practice in a nationally consistent way, which will ultimately be beneficial to all regulated entities.
During this financial year, I was also elected to be a member of the Main Commission of the International Commission on Radiological Protection (ICRP)¹, which allows me to provide further support to the development and national implementation of international best practice in radiation safety.
 ARPANSA's Fundamentals Document
ARPANSA's Fundamentals Document
Fundamentals
One example of alignment to the international framework for radiation safety is the publication this year of Fundamentals for Protection Against Ionising Radiation (RPS F-1). The Fundamentals builds on a number of high‑level international standards, recommendations, codes of conduct and conventions covering all applications of ionising radiation and all exposure situations, and integrates safety, security and radiation protection into one holistic, ‘fundamental’, approach to the management of radiation risks. The Fundamentals is a top level document in a revised hierarchy of fundamentals, codes and guides, intended to govern all uses of, and exposures to, radiation across all jurisdictions, developed by ARPANSA jointly with the states and territories through the Radiation Health Committee.
 A member of ARPANSA’s Dose Register Team at work
A member of ARPANSA’s Dose Register Team at workDose register
Another example of implementation of international best practice is the establishment of a database on occupational exposures of radiation workers, aligned with the requirements in the International Basic Safety Standards.² Based on an initial agreement with the then Department of Resources, Energy and Tourism, ARPANSA has developed the Australian National Radiation Dose Register. Originally, it was intended for uranium mine workers and has now, with the last mine coming on line this year, been implemented to all uranium mines licensed to operate in Australia.³ The register currently holds records of over 31 000 uranium mine workers. Work has commenced with the intent of expanding the register to other mining activities involving radioactive minerals, to Commonwealth licence holders, and beyond, as resources permit and obstacles can be overcome.
In relation to uranium mining, but also more generally in relation to exposure to radon and its progeny, the ICRP has advised that the dose conversion factors for some inhaled radioactive substances will change. This further emphasises the importance of better characterising the work environment in the mines. ARPANSA has carried out relevant measurements during the year, which will make it possible to further improve the understanding of the exposure of workers, and guide mitigation efforts. ARPANSA’s participation in the development of ICRP’s Publication 126 - Radiological Protection against Radon Exposure, has contributed to this objective.
Radiation protection of the patient
Of particular interest is the risks associated with the use of computed tomography (CT) scanning in diagnostic procedures involving children, which has been the subject of major studies internationally as well as in Australia (in which ARPANSA has participated). While the risk for cancer following such an examination is small and should not prevent justified medical examinations using ionising radiation, further fundamental research as well as guidance to the medical profession is warranted.
In October 2013, the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR), which I have the honour to chair, published its scientific assessment: Effects of radiation exposure of children.4 The UNSCEAR study considered 23 cancer forms and the relative sensitivity of children - compared to adults - in developing such cancers after exposure to ionising radiation. Children are clearly more likely than adults to develop some cancers after exposure to ionising radiation; examples of such cancers are leukaemia (except chronic lymphocytic leukaemia), thyroid cancer, breast cancer and non-melanoma skin cancer. For other cancer forms, the sensitivity seems to be about the same; for some cancer forms the relationship to ionising radiation is unclear for both children and adults, or data are inconclusive. The study provides a valuable summary of current knowledge as we go forward exploring the important issue of optimisation of radiation protection of the patient.
On this note, it is also worth pointing out that ARPANSA has entered into a Memorandum of Understanding with the Department of Health to develop and disseminate web-based information and educational material related to the use of ionising radiation for diagnostic purposes for the medical profession. ARPANSA also continues to survey data to support the development of diagnostic reference levels (DRLs) for a variety of diagnostic procedures. International experience demonstrates that implementation of DRLs tend to drive exposures downward, with no detriment to the diagnostic outcome. These activities occur against the backdrop of international guidance, noting also that exposure to ionising radiation in diagnostic procedures is now the major contributor to radiation exposure of the Australian population (see Figure 1).
Figure 1: Radiation exposure to the Australian population:
(Click for larger view)
There are three main sources of artificial ionising radiation. They are:
• medical uses, such as diagnosis
• industrial uses, mainly in measurement and
• scientific research natural background.
The chart shows the relative annual per capita dose to the Australian population from the various radiation sources.
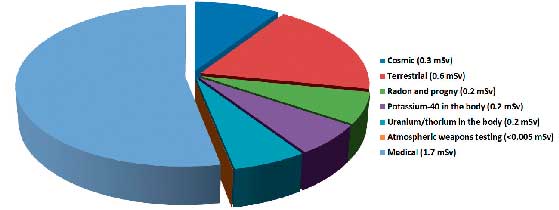 Radiation exposure to the Australian population
Radiation exposure to the Australian populationWorld Health Organization collaboration
ARPANSA’s collaboration with the World Health Organization (WHO) is formally recognised through ARPANSA’s status as a Collaborating Centre for Radiation Protection. WHO and the International Commission for Non-Ionizing Radiation Protection (ICNIRP) are key fora for development of protective approaches to non-ionising radiation, in particular, ultraviolet radiation (prevention of skin cancer) and electromagnetic radiation such as radiofrequency radiation used in mobile and fixed communication (an area where perceptions of health effects and risks are widely divergent, both among experts and among the general public).
ARPANSA supports the WHO’s work in both the ultraviolet radiation and electromagnetic radiation areas. An example of the importance of this collaboration is the opportunity it gives ARPANSA to tap into the most recent scientific evaluations at a time when ARPANSA is commencing work on review and revision of the radiation protection standards Maximum Exposure Levels to Radiofrequency Fields – 3 kHz to 300 GHz.5 During the year, ARPANSA published a review of recent studies of health effects of RF radiation6, which will form part of the underpinning scientific documentation supporting the revision.
As a member of the WHO Radiation Emergency Medical Preparedness and Assistance Network (with the Peter McCallum Cancer Institute), ARPANSA is sharing experience, information, best practices and technical expertise, in the public health and medical response to a radiation emergency.
The 2011 nuclear accident in Japan
The accident at the Fukushima Dai-ichi nuclear power station in 2011 has internationally prompted major reviews of safety practices among operators of nuclear facilities. The IAEA has reviewed its safety standards and is updating some of those standards with the participation of Member States. While Australia does not operate nuclear power reactors, ARPANSA has requested ANSTO consider Fukushima-related safety issues as relevant to safety of the Open Pool Australian Lightwater (OPAL) reactor. ARPANSA has also considered the first Periodic Safety Review of the OPAL reactor during the year. This material will inform considerations, in the beginning of next financial year, of the current OPAL operating licence and its associated conditions of licence.
In April 2014, UNSCEAR published its scientific assessment of the accident at Fukushima Dai-ichi nuclear power station: Levels and effects of radiation due to the nuclear accident following the great east-Japan earthquake and tsunami.7 A Japanese translation was published in May 2014. Whilst I chaired the Committee during the assessment, ARPANSA staff (through its Chief Radiation Health Scientist, Dr Stephen Solomon and others) led the international dose assessment team that estimated the radiation doses to different population groups.
Of particular relevance was the estimation of doses to the thyroid gland of infants and children, caused mainly by radioactive iodine released from the damaged reactors. The estimates of thyroid exposures of children after the accident carry significant uncertainties; however, the data indicates that the number of children exposed at levels where statistically discernible increases in thyroid cancer have been demonstrated in earlier studies, would be small. Overall, significant changes in health statistics, directly related to radiation exposure, are not expected. These findings do not negate that risks may have increased, nor disregard the suffering associated with any extra cancer case. Health statistics arising from the Fukushima Health Management Survey will only be possible to evaluate, with any degree of certainty, several years from now.
In addition, the suffering caused by evacuation, loss of livelihood and loss of family members through aggravation of health conditions that had been caused by the evacuation, is very significant among tens of thousands of people who remain evacuated in the wake of the nuclear accident. A further aggravating factor is the wide-ranging devastation of land, property and infrastructure caused by the effects of the earthquake and tsunami.
Major licence applications from ANSTO
During the year, I issued licences to prepare a site for, and to construct, two nuclear facilities at the Lucas Heights Science and Technology Centre (LHSTC) in Sydney’s south-west, namely the ANSTO Nuclear Medicine Molybdenum-99 Production Facility (the ANM Facility), and the Interim Waste Store (the IWS Facility). These decisions were made after careful review of the applications and the content of public submissions by ARPANSA staff from all operational branches (Regulatory Services, Radiation Health Services and Medical Radiation Services) and following discussions with, and advice from, the Nuclear Safety Committee.
The ANM Facility is intended to replace the existing radiopharmaceuticals production facility and increase the production capacity several-fold. The IWS Facility is intended to be an interim storage facility for radioactive waste returned from France in 2015. The waste originates from the reprocessing of fuel used for the operation of the now permanently shut down High Flux Australian Reactor, HIFAR. Australia is obliged to receive this waste by the end of 2015 at the latest. Whilst the waste was originally intended to be transported and stored at a planned National Radioactive Waste Management Facility, it is now clear that such facility will not be built in time to receive this waste. Any national facility will require the approval of the CEO of ARPANSA for siting, construction and operation.
During this financial year, ARPANSA also issued a licence to prepare a site for, and to construct, the Synroc Facility at the Lucas Heights Science and Technology Centre. This is a prescribed radiation facility intended to convert the liquid intermediate level waste resulting from the production of radiopharmaceuticals, to a ceramic form, using the ANSTO Synroc technique.
All documentation relevant to these licence decisions are available from ARPANSA’s website. A number of other licence applications from ANSTO and other entities were handled during the year. These are reported in further detail in subsequent sections of this Annual Report.
Status of radiation safety among licence holders
ARPANSA monitors compliance with the Australian Radiation Protection and Nuclear Safety Act 1998 (the Act), the Australian Radiation Protection and Nuclear Safety Regulations 1999 (the Regulations) and with any additional condition of licence imposed by ARPANSA, through the review of quarterly or annual reports submitted by licence holders, inspections, and other interaction with licence holders including a yearly event called the ‘Licence Holders’ Forum’.
During the year, ARPANSA declared four breaches with implications for safety and five with no or minor implications for safety among its licensees. The breaches were recorded in the Quarterly Reports by the CEO of ARPANSA to the Assistant Minister for Health and were tabled in Parliament. Safety breaches included: failing to follow the approved change control process and for use of an unauthorised tool (pry bar) during fuel assembly clamping at the OPAL Reactor; and, PETNET’s operation of a cyclotron without a functioning safety interlock system. The breaches are further detailed in Section 3.3 and Appendix 7, Table 13 of this Annual Report.
Incident reporting in Australia
ARPANSA maintains a nationwide incident register, called the Australian Radiation Incident Register (ARIR).8 During this financial year, ARPANSA reported four incidents to the register, including a contamination event resulting in a skin dose of 70% of the statutory limit (which was also a breach and is described in the Report on Performance). There was a total of 200 incidents for the 2013 calendar year. The majority of these incidents concern the medical uses of radiation, reflecting the high volume of medical radiation procedures conducted in Australia every year.
This number of incidents reported to the ARIR represents an increase from previous years, where the number of incidents over the last three years has been fairly stable at around 115. Analysis of this increase remains to be carried out; however, as a preliminary view, I would offer the hypothesis that this increase is at least partly due to proactive awareness-raising carried by ARPANSA, and which have resulted in an increased reporting of incidents to ARIR. Thus, the upward trend may be an illustration of improved safety culture rather than deterioration of safety per se.
In addition to the ARIR, ARPANSA is also responsible for reporting incidents to the IAEA International Nuclear Events Scale reporting scheme on behalf of Australia. There were no reportable events during the financial year 2013–14. Based on data in the ARIR and other information sources, such as inspections, the Australian National Radiation Dose Register and ARPANSA’s Personal Radiation Monitoring Service, and notwithstanding the occurrence of incidents and breaches, I conclude that I have no indication suggesting that radiation safety in Australia across jurisdictions and practices is generally deficient or unsatisfactory.
Regulatory governance
The Australian National Audit Office (ANAO) carried out a review of ARPANSA’s regulation of Commonwealth licence holders during the year.9 ANAO concluded that ARPANSA has been ‘generally effective in managing key aspects of the regulatory framework applying to the possession and use of radiation and nuclear sources and facilities by Australian Government entities’. It also notes that ‘overall, stakeholders reported that ARPANSA was approachable and professional, and commented on the generally positive working relationship between ARPANSA and its regulated entities’. However, the ANAO identified a number of areas for improvement and issued four major recommendations related to improved management of conflict of interest; and further strengthening of a risk-based approach to the regulatory activities. It also notes incomplete implementation of some of the earlier recommendations ANAO made.10
I have accepted the recommendations of the ANAO. Importantly, I take the observations in the audit report as support for the changes I have strived to implement over my tenure at ARPANSA as regards regulatory governance. I remain committed to implementing the recommendations, adapted as necessary to suit the approach to regulatory activities that I find most effective and proportionate, and report progress in the Annual Report. The way forward in relation to the main recommendations is outlined in Appendix 9.
These planned activities are well aligned with the Government’s deregulation agenda. A long-term program, captured under the umbrella ‘Regulatory Delivery Model’ has commenced which considers the proportionality (from a risk perspective) of ARPANSA’s regulatory activities with the aim of improving the efficacy of regulatory oversight and reducing regulatory burden, whilst not putting the safety outcome into jeopardy. I intend to include a more fulsome report on such activities and their impact on regulatory efficacy in the Annual Report covering the year 2014–2015.
As also pointed out by the ANAO, ARPANSA is not yet fully recovering the costs from the licence holders for its regulatory activities, although the situation has greatly improved in recent years. Parallel to the regulatory delivery model program, ARPANSA is also strengthening its framework for managing regulatory costs with the view to ensure, averaged over years, full cost recovery of its regulatory activities. ARPANSA has already received consent for adjustment of the fee structure for 2014–2015 to this effect.
Financial performance
ARPANSA delivered a small surplus of $1.0m over the year in part driven by effective management of its discretionary cost base.
Outlook
Creation of a leaner and more efficient Agency
Over the past few years, implementation of successive reductions in appropriation has been matched by a gradual reduction in staffing. By 30 June 2014, the staffing level was 132 FTE, and expected to go down to 124 in the beginning of the next financial year. This staffing number is down from the 152 FTEs as of 30 June 2011.
The reduction in staffing has taken place through a mix of planned down-sizing, natural attrition and voluntary redundancies. It has brought the Agency to a sustainable financial and staffing position. It was partly implemented through the Sustainable Futures Program that the Agency launched in to early 2014, in consultation with staff and their representatives pursuant to the provisions in the ARPANSA Agreement. This program not only addressed the staffing level but led to the implementation of 15 recommendations on adjustments in organisational structure and operations, as modified during the consultation.
This reduction has so far been accomplished with only minor adjustments of the Agency’s delivery – essentially all programs are still running. However, it is now necessary to consolidate the position for the future. Continued efforts involve prioritisation (an improved planning process has contributed to this), succession planning, sustainable radiation protection services (including a major reinvestment and modernisation program), creating strategic relationships (through cooperation arrangements nationally and internationally governed by a national and an international engagement strategy), and general vigilance of discretionary expenditure. At the end of the financial year there had been no decision regarding future funding of the Australian Clinical Dosimetry Service.
A further challenge is to adapt the staffing profile to the needs. This will naturally also consider recruitment of specialised competence, noting the specialised nature of the Agency’s activities.
While ARPANSA has dealt with a number of major licence applications during the year, it is expected that the pressure will be high on the regulatory staff next year as well. The regulatory delivery model and cost recovery projects will eventually help in streamlining the regulatory activities and contributing to the deregulation agenda. Major outcomes will be improved regulatory service focusing on the essentials, but with unchanged ambitions of protection of health and the environment.
The demand on ARPANSA staff to provide advice to government, other agencies, and the general public, is expected to remain high. Again, the ambition is to further streamline processes and engage in a constructive way with stakeholders.
ARPANSA, through its staff, experience and competence, is well placed to take on the challenges in the future – although I am also certain this will necessitate changes, adaptation and decisions that may not always be easy.
Sustainable Futures 2014ARPANSA’s Strategic Management Committee released its Sustainable Futures 2014 and Beyond strategy in January 2014. The strategy focuses on developing an organisation that meets the needs of Government whilst maintaining the Agency’s financial position from 2014 and beyond. The Sustainable Futures program ensures that a more co-ordinated and strategic approach is applied to the establishment of relationships with ‘sister organisations’ nationally and world-wide via Memoranda of Understanding. Additionally, Sustainable Futures aims to ensure that services provided internally and externally are delivered by the best possible service delivery models. In keeping with the Agency’s Strategic Directions for 2012-16, ARPANSA will continue to develop its leaders and its workforce. ARPANSA is committed to building a high performance organisation and aligning its human capital management strategies with the Agency’s organisational direction. |
1. The ICRP Main Commission has thirteen members and is chaired by Dr Claire Cousins, UK. More than 200 radiation experts, regulators and policy makers are involved in ICRP’s work, which is organised in five Committees covering: biological effects (chaired by William F Morgan, USA); dosimetry (chaired by John Harrison, UK); medical radiation (chaired by Eliséo Vañó, Spain); application (chaired by Jacques Lochard, France), and environment (chaired by Carl-Magnus Larsson, Australia).
2. Radiation Protection and Safety of Radiation Sources: International Basic Safety Standards, GSR Part 3, IAEA, 2014. Normally referred to as the BSS; it is published by the IAEA on behalf of its member states and is co-sponsored by the European Commission, the Food and Agriculture Organization of the United Nations, the International Atomic Energy Agency, the International Labour Organization, the Nuclear Energy Agency of the Organization for Economic Cooperation and Development, the Pan American Health Organization, the United Nations Environment Programme, and the World Health Organization.
3. These are: Olympic Dam, Beverley and Honeymoon (care and maintenance) in South Australia; and Ranger in Northern Territory.
4. UNSCEAR 2013 Report to the UN General Assembly (A/68/46): Volume II: Sources, effects and risks of ionizing radiation. Annex B - Effects of radiation exposure of children.
5. Radiation Protection Series No. 3, ARPANSA 2002.
6. Report by the ARPANSA Radiofrequency Expert Panel: Review of Radiofrequency Health Effects Research – Scientific Literature 2000 – 2012’ ARPANSA Technical Report Series No. 164 at www.arpansa.gov.au/pubs/technicalreports/tr164.pdf.
7. United Nations Scientific Committee on the Effects of Atomic Radiation, (April 2014) Annex A - Levels and effects of radiation exposure due to the nuclear accident after the 2011 great east-Japan earthquake and tsunami.
8. The definition of incidents for the purpose of the ARIR is outlined in Schedule 13 of the National Directory for Radiation Protection, Radiation Protections Series 6: www.arpansa.gov.au/RadiationProtection/arir
9. Australian National Audit Office: Regulation of Commonwealth Radiation and Nuclear Activities, Performance Audit Report No. 29 2013–14.
10. Australian National Audit Office: Regulation of Commonwealth Radiation and Nuclear Activities, Audit Report No. 30 2004-05.


