Part 3: Report on Performance - 3.3 Ensure effective regulation and enforcement activities
This Page:
- Major achievements
- Challenges
- Major licensing activities
- Other significant activities in relation to regulatory oversight
- Inspections
- Breaches
- Transport of radioactive material
- Incidents
- ANAO performance audit
- National uniformity
During this financial year, ARPANSA's licensing and compliance workload was dominated by the assessment of major licence applications for new ANSTO facilities including the interim waste store facility, a new and expanded molybdenum-99 manufacturing facility and the waste treatment facility using ANSTO's Synroc technology, known as the 'SyMo' facility.
Major achievements
- Evaluating lengthy and complex siting and construction licence assessments for three major controlled facilities at ANSTO.
- Further work on ARPANSA's holistic safety practices.
- Conducting 42 inspections and site visits – exceeding the target of 35.
- Publication of top-tier radiation protection document - Radiation Protection Series F-1 Fundamentals for Protection against Ionising Radiation.
- Obtaining 'in-principle' agreement from ARPANSA's Council and Committees, to use IAEA Safety Standards publications as the basis for International Best Practice in Radiation Protection for the purposes of developing Nationally Uniform guidance for the Australian situation.
Challenges
- Communicating to licence holders the benefits of holistic safety practices.
- Achieving national uniformity in radiation protection and nuclear safety.
Major licensing activities
During the financial year, ARPANSA issued the following new or amended facility licences as follows:
- Nuclear installation licence (F0270) to prepare a site for the ANSTO Nuclear Medicine Molybdenum-99 Facility at Lucas Heights Science and Technology Centre issued to ANSTO on 4 October 2013.
- Prescribed radiation facility licence (F0280) to operate the 1MV Compact Accelerator within the Centre for Accelerator Science at Lucas Heights Science and Technology Centre issued to ANSTO on 9 October 2013.
- Amended prescribed radiation facility licence issued to the Director of National Parks on 27 November 2013 establishing dose constraints for the South Alligator Disposal Facility.
- Nuclear installation licences to prepare a site (F0277) and to construct (F0279) the Interim Waste Store at Lucas Heights Science and Technology centre issued to ANSTO on 29 November 2013.
- Amended prescribed facility licence (F0271) issued to Synchrotron Light Source Australia Pty Ltd with revised licence conditions on 20 December 2013.
- Amended prescribed radiation facility licence (F0280) issued to ANSTO to undertake routine operations of the 1 MV Compact Accelerator within the Centre for Accelerator Science at Lucas Heights Science and Technology Centre on 28 April 2014.
- Prescribed radiation facility licence (F0266) to prepare a site for and construct the ANSTO SyMo facility at Lucas Heights Science and Technology Centre issued to ANSTO on 13 May 2014.
- Prescribed radiation facility licence (F0287) to construct the 6 MV accelerator known as SIRIUS at Lucas Heights Science and Technology Centre was issued to ANSTO on 29 May 2014.
- Nuclear installation licence (F0285) to construct the ANSTO Nuclear Medicine Molybdenum-99 Facility at Lucas Heights Science and Technology Centre was issued to ANSTO on 27 June 2014.
Other significant activities in relation to regulatory oversight
- On 16 November 2013, ARPANSA granted approval to ANSTO under Regulation 51 to make a change in shift roster at ANSTO Health.
- On 12 December 2013, ARPANSA and ANSTO met to progress the OPAL Research Reactor Periodic Safety Review which is expected to be completed by the end of the first quarter of the 2014–15 financial year.
- On 13 January 2014, ARPANSA granted approval to ANSTO under Regulation 51 requests for approval to:
- change in inventory limits at ANSTO Health facilities
- the hot commissioning and operation of Lu-177 hot cells
- On 11 April 2014, ARPANSA granted ANSTO permission under Regulation 51 to undertake an additional molybdenum-99 production run per week at Building 54 up to 8 times per year.
- During the financial year, ARPANSA also issued amended source licences to hot commission the ANSTO Bragg Institute neutron beam instruments JOEY, KOOKABURRA, DINGO and BILBY. In addition, amended source licences have been issued to undertake routine operations at KOOKABURRA and JOEY.
- ARPANSA undertook the first of a series of planned thematic inspections of its licence holders. The first thematic inspection involved the delivery of radiation protection services across the ANSTO organisation, with a view to identifying holistic safety characteristics and attitudes across ARPANSA licence holders.
Inspections
During the course of the year ARPANSA undertook a planned inspection program of sources, prescribed radiation facilities and nuclear installations operated by licence holders in order to monitor compliance with the ARPANS Act and the ARPANS Regulations.
A summary report of these inspections can be found on the ARPANSA website at: www.arpansa.gov.au/Regulation/Inspections
The inspection program was planned on the basis of:
- licence holder risk ranking
- licence holder compliance history
- licence holder incident and accident history, and
- date of last inspection.
Breaches
Breaches with safety implications
Four breaches with potential safety implications were identified during the financial year as follows:
- PETNET Australia – breach of Regulation 49 by operating the PETNET cyclotron without a functioning safety interlock system.
- PETNET Australia – breach of Regulation 51 by implementing a Safe Operating Procedure with significant implications for safety without the appropriate approval.
- ANSTO OPAL Reactor – breach of Regulation 49 by failing to follow the approved change control process for use of an unauthorised tool during fuel assembly clamping.
- ANSTO Life Sciences – breach of Regulation 49 by not following its quality control procedures and change control process.
In relation to the two breaches by PETNET Australia, the licence holder (PETNET) accepted the conclusions of ARPANSA's inspection report and the licence holder returned to compliance with no formal enforcement action had to be pursued by ARPANSA.
In relation to ANSTO OPAL Reactor breach of Regulation 49, ANSTO returned to compliance by making appropriate redesign changes and no formal enforcement action had to be pursued by ARPANSA.
In relation to the ANSTO Life Sciences breach of Regulation 49, ANSTO implemented appropriate corrective actions to return to compliance and no formal enforcement action was pursued by ARPANSA.
Breaches with no or minor safety implications
During the financial year, breaches with minor or no safety implications were recorded across the following areas:
- Disposal of controlled apparatus without prior approval from ARPANSA. The equipment was returned to an x-ray equipment supplier who is appropriately licensed to deal with the devices.
- Failure to follow own safety procedures.
- Possession of controlled apparatus without an appropriate licence in place. An application was subsequently submitted and the appropriate licence granted.
- Failure to undertake radiation monitoring surveys around controlled apparatus at the recommended frequency.
- Possession of a controlled apparatus without the appropriate licence in place. However the controlled equipment was not used by the licence holder until an appropriate licence was obtained.
- Exceeding a building activity limit with no safety implications.
In all of the above cases, the breach was assessed to have minor safety implications, corrective actions were implemented by the licence holder and no enforcement action was considered necessary.
During this financial year, 42 inspections and site visits were achieved against a target of 35.
ARPANSA is committed to carrying out its regulatory functions in a responsive and timely manner. In relation to timeliness of assessing licence applications we advise that:
- On average, Regulation 51 requests for approval were processed in 18 days against a target of 30 days
- On average source licence applications were processed in 43 days against a target of 30 days
- On average facility licence applications for large facilities were processed in 96 days against a target of 60 days.
The target for source and facility licence applications was not met due to the complexity of new applications for nuclear installations and prescribed radiation facilities received from ANSTO. As a result, ARPANSA is working with large licence holders to improve the forecasting and management of upcoming licensing requirements. The Key Performance Indicator for inspections was reduced from last financial year in acknowledgement of the resource burden of a forecast number of applications for ARPANSA to manage.
Performance against key performance indicators
| Quantitative Indicators suggested by Australian National Audit Office (ANAO) | 2012–13 Revised Budget | 2013–14 Achieved |
|---|---|---|
| Timeliness of Licence Application Assessments | ||
| Average time to assess facility licence applications |
<60 days
|
96 days
|
| Average time to assess source licence applications |
<30 days
|
43 days
|
| Average time to assess Regulation 51 requests |
<30 days
|
18 days
|
Qualitative Deliverables for Program 1.1
| Quantitative Deliverables | 2012–13 Revised Budget | 2013–14 Budget Target | 2014–15 Forward Year 1 | 2015–16 Forward Year 2 | 2016–17 Forward Year 3 |
|---|---|---|---|---|---|
| Ensure effective regulation and enforcement activities | |||||
| Number of inspections and site visits of facilities holding a Commonwealth licence | 60 | 35 | 35 | 35 | 35 |
| MET | 59 | 42 | N/A | N/A | N/A |
| CASE STUDY: Licensing of Nuclear Installations and Radiation Facilities |
|---|
|
During this financial year, ARPANSA licensed ANSTO to operate three significant nuclear installations and radiation facilities: ANSTO Nuclear Medicine Molybdenum-99 (ANM Mo-99) Facility, Interim Waste Store (IWS) and ANSTO's SyMo Facility. ANM Mo-99 Facility In October 2013, ARPANSA issued a licence (F0270) to ANSTO to prepare a site for the ANM Mo-99 Facility and in June 2014, a licence (F0285) for construction of this facility. The proposed facility will be used to produce molybdenum-99 (Mo-99), the precursor of technetium 99m (Tc-99m) which is the radionuclide used in about 80% of all diagnostic nuclear medicine procedures for diagnosing various diseases such as heart and bone diseases. Apart from meeting the demand of the domestic Australian market for Tc-99m, this facility will also have the capacity to export which will help secure the future global supply. The proposed facility will have capability of producing Mo-99 at about four times of the current level. Molybdenum-99 will be produced by neutron irradiation of low enriched uranium (LEU) at OPAL reactor. The use of LEU will enhance the international efforts for nuclear non-proliferation. In granting licences to ANSTO, ARPANSA's assessment has taken into account relevant legislative requirements, design and technological aspects including the increased level of production, current use of similar technology in other countries, operating experience in a similar process and use of improved technology in controlling airborne emissions, contents of public submissions, operational waste contingencies, decommissioning and management of decommissioned wastes at the end of facility life and the contents of public submissions. The ANM Mo-99 Facility is currently under construction. Intermediate Waste Store In November 2013, ARPANSA issued a licence (F0277) to ANSTO to prepare a site for the Intermediate Waste Store, and a licence (F0279) for construction of this facility. The IWS Facility will store radioactive waste resulting from reprocessing of fuel that was used in the now permanently shut down High Flux Australian Reactor (HIFAR). This includes residual fission products after separation of fissile material from the fuel and secondary waste resulting from the reprocessing operations; the secondary waste, referred to as ‘technological waste', includes mechanical components such as piping, valves, pumps and protective clothing such as gloves that have been contaminated during the reprocessing. This waste will be immobilised in a vitreous form and transported/stored in an engineered shielded dual storage and transport container known as the TN81. In addition, technological waste will be returned from France cemented within steel drums. The IWS may also be required to store radioactive waste returning from the United Kingdom, which is due to return around 2020, if the National Radioactive Waste Management Facility is not available at that time. In granting a licence to site and construct this facility, ARPANSA's assessment has considered relevant legislative requirements, design features, international best practice, contingencies and contents of public submissions. The IWS Facility is currently under construction. SyMo Facility In May 2014, ARPANSA issued the licence (F0266) to ANSTO to site and construct a purpose built intermediate level liquid waste conditioning facility, namely the SyMo Facility. The proposed SyMo facility will apply Synroc (synthetic rock) technology, developed by ANSTO, for immobilisation of intermediate liquid waste generated from Mo-99 production process into highly immobilised glass ceramic and/or ceramic form for the safe disposal of radioactive waste. The facility is designed to manage current and future alkaline intermediate level liquid waste, from ANM Mo-99 Facility and acidic legacy waste. Regulation 40 requires the CEO to invite public submission if the facility is a nuclear installation. Though the proposed facility is a prescribed radiation facility, public submissions on this facility were invited to consider its interface with the ANM Mo-99 Facility. In granting a licence to site and construct the proposed SyMo Facility, ARPANSA's assessment has taken into account relevant legislative requirements, conservative proven design and engineering practice and contents of public submissions. In addition, ARPANSA also requires ANSTO to undertake a full scale trial using non-radioactive material as part of testing and commissioning. |
Figure 6: Licensing phases
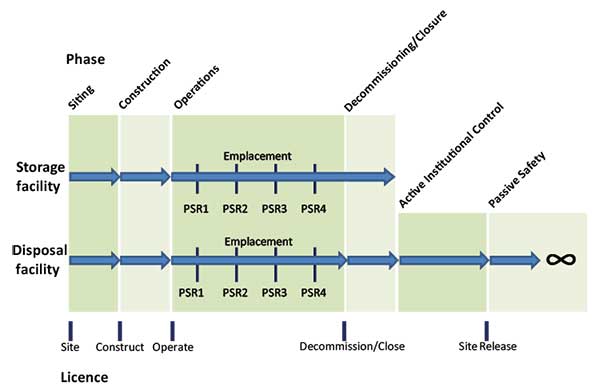
Diagrammatic representation of the licensing phases for a radioactive waste store (top) and radioactive waste disposal facility (below). PSR refers to Periodic Safety Review
Transport of radioactive material
During this financial year ARPANSA issued one certificate of design, one certificate of approval and validated five package designs for the transport of radioactive material.
ARPANSA approved the package design of model 1860A and issued the following certificate of design to Analogue and Digital Measurements Pty Limited, Australia:
- AUS/2013-47/B(U)-96
ARPANSA validated five overseas Package Designs and issued the following certificates of validation to ANSTO:
- AUS/2013-48/B(U)F-96
- AUS/2013-49/B(U)-96
- AUS/2014-50/B(U)F-96
- AUS/2014-51/B(U)-96
- AUS/2014-52/B(U)-96
ARPANSA also issued the following certificate of approval of zirconium-89 radionuclide value to Sir Charles Gairdner Hospital:
- AUS/2014-53/Zr-89/RV-96.
Incidents
The following three safety incidents among Commonwealth licence holders were reported for this financial year:
ANSTO Bragg Institute
On 27 August 2013 work was being undertaken on the installation of a new neutron beam experiment fitted to a previously unused beam line HB-2. An area radiation monitor alarmed in the Reactor Beam Hall and a single worker in the area at the time evacuated. The alarm was also registered by the Reactor Control and Monitoring System and operators followed standard management practices to investigate the cause. The investigation identified that adjacent primary shutter (HB-1), had accidentally opened. The shutters are used to block the passage of neutrons from the reactor core to neutron beam apparatus when not needed. The shutter concerned was an unused beam and was fitted with additional concrete shielding and the low level of radiation measured was the result of neutron scatter and not the direct beam. The maximum dose to any worker was assessed as negligible (1 microsievert). An investigation into the incident discovered that it was caused by the shutter control circuits incorrectly interpreting electrical interference, from the closing of the HB-2 shutter, as a command to open the unused HB-1 shutter. The design of HB-1 and HB-2 control circuitry differs from other shutter controls and a number of design weaknesses were identified and modifications were introduced to prevent this from reoccurring. In the interim period before these modifications, these particular shutters were electrically isolated to prevent them from opening. ARPANSA was satisfied with the ANSTO safety response to this incident and with the adequacy of modifications made.
ANSTO Life Sciences – Camperdown Sydney
On 30 October 2013, a worker at ANSTO Life Sciences' Camperdown Facility was contaminated with a liquid radioactive source, fluorine-18 while handling a syringe containing the quality control sample, contaminating the hands of the worker, some clothing and personal items. ANSTO's analysis of the dose implications from the incident estimated that the worker received a dose of 355 millisieverts to the hands which is 70 percent of the annual statutory dose limit (500 mSv to the hands and feet). ANSTO assessed the incident as a near miss of a 7.1 sievert dose. An internal ANSTO investigation into the cause of the incident was undertaken. ARPANSA also undertook its own review and inspection of the incident. A number of causes identified included weaknesses in risk assessment, failure to work within approved operating limits and poor safety culture. ARPANSA made several recommendations to ANSTO to improve work practices and ARPANSA is monitoring implementation. ANSTO was also found to have breached Section 30(2) by failing to comply with Regulation 49 by not following its own plans and arrangements for managing safety.
ANSTO – OPAL – tellurium rig dropped into reflector vessel
On 3 June 2013, during the transfer of a tellurium rig from the OPAL irradiation facility in the reflector vessel, the rig detached from the lifting tool and fell from approximately one metre under water onto the reflector vessel. Operators had verified that the rig nozzle was appropriately latched to the lifting tool in accordance with safety requirements prior to removing the rig from the facility. The rig was raised from the reflector vessel facility and the operators commenced transferring the rig to the Service Pool. Immediately following the incident, the Utilisation Manager and Reactor Manager were notified, the risk was assessed and the recovery plan undertaken. After a period of two hours, the rig was recovered and transferred to the Service Pool.
Following the incident, both the rig and reflector vessel were inspected for damage using the underwater camera but no deformation was observed. The reflector vessel is designed to withstand impact of a 100 kilogram silicon ingot which is significantly greater than the impact of the 5 kilogram tellurium rig. The rig was visually inspected in the hot cell, found to be fully operational and returned to service. The tellurium targets were unloaded from the rig undamaged.
No abnormal radiation doses were received by the personnel involved and no release of radioactive material into the pool water or air was detected at any time during the incident. ARPANSA was advised of this incident during their 5 June 2013 inspection. The event is still under ANSTO's investigation. The event causes have not been confirmed.
None of the incidents listed above qualified for the IAEA's International Nuclear and Radiological Event event rating Scale as the safety significance of all of these incidents was well below their lowest designated level of anomaly.
Performance against key performance indicators
| Qualitative Deliverables | 2012–13 Revised Budget | 2013–14 Budget Target | 2014–15 Forward Year 1 | 2015–16 Forward Year 2 | 2016–17 Forward Year 3 |
|---|---|---|---|---|---|
| Ensure effective regulation and enforcement activities | |||||
|
Number of safety incidents involving Commonwealth users of radiation |
<10 | <10 | <10 | <10 | <10 |
| MET | 6 | 3 | N/A | N/A | N/A |
ANAO performance audit
In May 2014, the Australian National Audit Office (ANAO) released their report into Regulation of Commonwealth Radiation and Nuclear Activities that commenced in April 2013. The findings of that report are described in detail in the External Scrutiny and Advice.
National uniformity
ARPANSA promotes national uniformity and international best practice in radiation and nuclear safety through the National Directory for Radiation Protection (the National Directory) which is jointly developed by ARPANSA and state and territory radiation regulators through the Radiation Health Committee.
After endorsement by the Radiation Health Committee, the Radiation Health and Safety Advisory Council (the Council) and consideration by the Office of Best Practice Regulation (OBPR), in February 2014 ARPANSA published Radiation Protection Series F-1 Fundamentals for Protection Against Ionising Radiation. This publication is the top tier document in the Australian national framework to manage risks from ionising radiation.
The proposed sixth amendment of the National Directory to Schedules 5, 6, 9 and 13 (covering a number of topics including: expansion of the licensing criteria for chiropractors; clarification of the incident reporting requirements and editorial matters) was considered out of session by the Standing Committee on Health (SCoH) following endorsement by the Australian Health Ministers' Advisory Council. The SCoH endorsed the sixth amendment, and the amended National Directory was published in February this year. However, work continues on a more significant amendment to Schedule 13 National Incident reporting.
Work continued on a proposed amendment of the National Directory relating to Personal Radiation Monitoring Service provider certification. The Radiation Health Committee considered that, as an interim measure, Australian jurisdictions could apply the guidance of chapter 7 of the International Atomic Energy Agency's (IAEA) draft safety guide DS453 Occupational Radiation Exposure.
The Radiation Health Committee considered a draft statement Regulatory Expectations for Users of Radiation Sources Seeking to Obtain Authorisations in More than One State or Territory. Once finalised, this statement should provide a mechanism for reducing unnecessary regulatory and additional costs for business within existing agreed national policy of mutual recognition.
Work continued on a proposed amendment of the National Directory on user disposal of very low-level radioactive material intended to replace the National Health and Medical Research Council's Code of practice for the disposal of radioactive waste by the user (1985). Work continued on a proposed amendment of the National Directory for control of intense pulsed light (IPL) and lasers for cosmetic use, and the Radiation Health Committee agreed to proceed to a Consultation Regulatory Impact Statement (in accordance with the requirements of the OPBR).
The Radiation Health Committee agreed that the Radiation Protection Series should, over time, reflect the IAEA's framework and hierarchy of radiation protection publications. The Radiation Health Committee also considered adopting, where possible and with regard to the Australian context, IAEA and other international best practice publications as a means of revising and updating the Radiation Protection Series. It was recognised that, given the limited resources available within all jurisdictions, this approach would be an effective and appropriate means of providing nationally uniform Australian radiation protection requirements and guidance.
A formal review of the National Directory in 2007 found that while it has contributed to national uniformity, there is room for improvement. In June 2008 the Radiation Health and Safety Advisory Council concluded, among other things, that the National Directory model should be reviewed at an appropriate time to ensure that more efficient regulatory processes for improving national uniformity can be developed. ARPANSA, through the Radiation Health Committee, has commenced work on developing options to improve national uniformity in radiation protection policies and practices.
| CASE STUDY: ARPANSA's Holistic Safety Program |
|---|
ARPANSA has developed and promotes a holistic approach to radiation safety in its regulatory functions. Holistic safety refers to a best practice approach to safety management which considers the interaction and interdependence between three key aspects relevant to the handling of nuclear and radiological materials, namely: human, technological and organisational aspects to both create and maintain safe operations and reduce the relative risk. Research into accident causation found that the promotion of holistic safety can empower licence holders to improve safety practices by providing both the operator and the regulator with a deeper understanding of the dynamic factors affecting nuclear and radiological safety. ARPANSA's holistic safety program supports a risk-informed approach to regulation in dealing with licence holders. A holistic safety program is not expected to result in an increased regulatory burden to licence holders who are currently operating consistently and in accordance with Holistic Safety principles. Where an assessment of issues from a Holistic Safety perspective raises new aspects that may impact on safety, these issues will be managed as part of the normal practice in current inspections. This holistic approach (also called systemic safety) is increasingly being adopted by international organisations (such as the International Atomic Energy Agency) and is being favourably considered by many businesses, regulators and organisations around the world. Holistic Safety principles have developed from the analysis and lessons learned from incidents, accidents, and real-life events. ARPANSA has taken these principles to form the basis of its Holistic Safety Guidelines (www.arpansa.gov.au/Regulation/Holistic/HolisticSafetyGuidelines.cfm). Holistic safety is structured around seven key characteristics relevant when analysing the inter-relationships between individuals, technology and organisational factors in a nuclear or radiological setting. These characteristics are: human factors; non-technical skills; resilience; defence in depth; integrated safety management; safety culture; and, protective security and security culture. ARPANSA's Holistic Safety Guide is an enabling document designed to encourage and assist licence holders to review their operations and is also used by ARPANSA's regulatory staff in assessments and inspections. The guide describes the attributes of each characteristic and is accompanied by a second publication with more specific questions which explore how an organisation performs against each characteristic. It is the overlap between the characteristics that helps to build an understanding of the interactions between individual, technological and organisational factors. ARPANSA has begun to undertake holistic safety assessments of licence holders. The objective is to map our understanding of safety strengths and vulnerabilities of a licence holder. This information is used to engage with the licence holder on safety management, to make comparisons between organisations to promote cross organisational learning, and to make improvements to ARPANSA's risk informed inspection program. ARPANSA has also developed micro and macro tools in the form of questionnaires available on our website to assist licence holders to undertake their own holistic assessments. ARPANSA cooperates with the international community by sharing its experience and knowledge of our holistic approach to safety. This also helps to keep ARPANSA at the leading edge of developments on the subject. |
16. Absorbed dose is the energy absorbed per unit of mass by ionising radiation.
17. Air kerma in air is the sum of kinetic energy of all charged (ionised) particles liberated per unit of mass.
18. Picard, S, Burns, D, Roger, P, Harty, P, Ramanathan, G, Lye, J, Wright, T, Butler, D, Cole, A, Oliver, C, and Webb, D, 'Key comparison BIPM.RI(I)-K6 of the standards for absorbed dose to water of the ARPANSA, Australia and the BIPM in accelerator photon beams', Metrologia 51 Tech. Suppl. 06006 (2014).
19. Ramanathan, G, Harty, P, Wright, T, Lye, J, Butler, D, Webb, D and Huntley, R. The Australian Primary Standard for absorbed dose to water (graphite calorimeter) (2014) 2013, Technical Report Series No. 166, June 2014.


