Part 1: Review by the CEO – Dr Carl-Magnus Larsson
This Page:
- International and National Perspective

- Performance Against Corporate Key Performance Indicators
- Regulation
- Science
- Service
- Finance
- Staff - Outlook for 2013-14
I am pleased to present to you the fifteenth Annual Report of the CEO of ARPANSA, the fourth under my stewardship of the Agency. In this review, I present highlights in radiation protection and nuclear safety during the year, as well as some corporate developments.
International and National Perspective
Even though many months have elapsed since the nuclear accident at the Fukushima Dai-ichi nuclear power plant in Japan, the accident continues to be a major consideration in international and national efforts to enhance nuclear safety, and assessments of the health implications are ongoing. Even at the end of the year, radioactive substances continued to leak from the site to the marine environment. Workers on site are tasked with complex operations including preventing further leaks, decontamination of the site, removal of fuel from the storage pools adjacent to the reactors, and the planning and subsequent execution of dismantling works. A very large number of people, previously living close to the power plant and in the so-called deliberate evacuation zone extending to the north-west of the facility, remain displaced. The nuclear accident has aggravated the already considerable human and societal toll taken by the Great East-Japan Earthquake and Tsunami, which includes the loss of approximately 20 000 lives as a direct consequence of the natural disaster.
The Government of Japan jointly with the International Atomic Energy Agency (IAEA) organised a Ministerial Conference on nuclear safety, held in Koriyama, Fukushima Prefecture, in December 2012. I had the honour of leading the Australian delegation to the Conference. The Australian statement emphasised the need for implementation of the internationally agreed safety requirements and for further enhancing transparency and information exchange within the international peer review system.
The health implications of the accident have been the subject of a study by the World Health Organization (WHO), published early in 2013. The study was based on estimates of radiation doses, published in May 2012, on the basis of information available in the first few months after the accident. The United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR), which I Chair, has, on the basis of a much expanded database and longer follow-up time, arrived at broadly similar estimates of doses compared to the WHO data. It seems that the prevailing weather conditions and early implementation of countermeasures have limited radiation doses to the population. While it is important to remain cautious in drawing conclusions, due to large inherent uncertainties in the estimations, it now appears that the direct impact of radiation on the health of the population will be limited. However, the impact on social and mental wellbeing is substantial. In conclusion, there is never any room for complacency and the vigilance on safety needs to be continued to further reduce likelihood and consequences of major accidents, whilst accepting that such accidents indeed are possible.
The IAEA is performing a comprehensive study of the accident, at the request of the IAEA Director-General Amano. The report will likely shed further light on the technical aspects of the accident. ARPANSA leads the working group on post-accident assessment. The report is planned to be finalised and delivered to Director-General Amano in 2014.
 (From left to right:) Mr Denis Flory, IAEA Deputy Director General for Nuclear Safety and Security, Dr Carl-Magnus Larsson, CEO of ARPANSA, and Mr Greg Webb, IAEA Press and Public Information Officer, talking to the press after the week-long International Experts' Meeting on Decommissioning and Remediation After a Nuclear Accident. IAEA Headquarters, Vienna, Austria, 1 February, 2013 (Photo Credit: Ayhan Evrensel/IAEA)
(From left to right:) Mr Denis Flory, IAEA Deputy Director General for Nuclear Safety and Security, Dr Carl-Magnus Larsson, CEO of ARPANSA, and Mr Greg Webb, IAEA Press and Public Information Officer, talking to the press after the week-long International Experts' Meeting on Decommissioning and Remediation After a Nuclear Accident. IAEA Headquarters, Vienna, Austria, 1 February, 2013 (Photo Credit: Ayhan Evrensel/IAEA)While the accident has had significant impact in Japan and internationally, in particular, in nuclear power countries, there have also been a number of significant developments nationally such as major investments into expanded radiopharmaceuticals production at the Australian Nuclear Science and Technology Organisation (ANSTO) Lucas Heights site in Sydney's south-west. As a result of these, ARPANSA has received an application from ANSTO to prepare a site for a facility for production of molybdenum-99 (a precursor of technetium-99 used in nuclear medicine procedures), which will four-fold increase the production capacity at the site. The liquid waste from the production is planned to be treated and solidified using the Synroc (synthetic rock) technique, which if the plans go ahead will be the first large-scale utilisation of this technique. ARPANSA is currently reviewing an application from ANSTO to prepare a site for, and construct, a facility built for this purpose, the SyMo facility. I expect that I will be able to make determinations in relation to both applications early next financial year.
I have also received applications to prepare a site for, and to construct, an interim radioactive waste storage facility to be built at the ANSTO Lucas Heights site, which is intended to receive the waste resulting from the reprocessing of HIFAR1 fuel in France and that will be returned from France in 2015, and potentially also for storage of similar type of waste returned from the United Kingdom around 2020. The need for an interim storage facility stems from the absence, at this point in time and in at least the next few years, of a national radioactive waste management facility. The technical review of the proposed interim storage facility was in an advanced stage at the end of the financial year. I expect to make a determination early in the next financial year.
Both the waste store and the radiopharmaceuticals production facility have been the subject of public consultation. The SyMo facility was included in the consultation because of its operational linkage to radiopharmaceuticals production, although public consultation was not legally required.
A number of other developments have occurred over the year. These include the issuing of an ARPANSA licence to operate the Australian Synchrotron in Melbourne (previously operating under a licence from the Victorian authorities) and a number of other activities in the regulatory, medical and scientific areas, with regard to both ionising and non-ionising radiation. These are covered in Report on Performance later in this Annual Report.
Performance Against Corporate Key Performance Indicators
ARPANSA's activities are planned and executed within ten different Key Areas identified in the ARPANSA Strategic Directions 2012-2016 (which are aligned with the obligations under the Australian Radiation Protection and Nuclear Safety Act 1998 and other acts governing the Agency's activities (see: www.arpansa.gov.au/AboutUs/Corporate/strategic.cfm).
ARPANSA's Corporate Plan for 2012–2013, defines a number of Corporate Key Performance Indicators. These, together with the Portfolio Budget Statements, form a framework of benchmarks against which I can monitor progress in the Agency's delivery of radiation protection and nuclear safety to the Australian Government and community.
Progress against the Portfolio Budget Statements and Agency Business Plan are described in the Report on Performance section of this Annual Report.
REGULATION
Number of licensee incidents – less than 10 per annum
ARPANSA maintains regulatory oversight over 42 licensees, holding 33 facility licences and 61 source licences. The actual number of sources covered by the licences is in the order of tens of thousands. Table 17 in Appendix 7 provides further details. Among the licensees are a number of large organisations, including ANSTO, the Commonwealth Science and Industrial Research Organisation (CSIRO) and the Department of Defence. On the other end of the spectrum are a number of small licensees with only minimal use of radiation in their operations.
The definition of an incident for various uses of radiation is spelled out in the National Directory for Radiation Protection (Radiation Protection Series No 6). In the 2012–13 financial year there were six reports of incidents among Commonwealth entities holding an ARPANSA licence. This is consistent with the two preceding years (five in 2011–12; and four in 2010–11).
The safety of the operations is verified through quarterly and annual reports to ARPANSA and by means of the Agency's inspection program. As for the previous year, ARPANSA essentially met its target of sixty inspections and site visits in a year (fifty-nine were undertaken in 2012–13). During this financial year, most inspection reports have been made publicly available on the ARPANSA website to foster transparency and to improve safety accountability among licence holders as well as within ARPANSA itself.
Despite efforts among licence holders and ARPANSA's surveillance, five breaches with safety significance have been reported in the Quarterly Reports that are tabled in Parliament. Eight breaches with no or small safety significance were recorded as well. Corrective actions have been taken. A number of potential breaches were still under investigation at the end of the year.
Taken collectively, and notwithstanding the fact that breaches with safety significance have been identified, the information available to ARPANSA provides me with reasonable reassurance that the operations of licence holders are over-all safe.
Enhancement of the safety culture among licensees will be aided by the Holistic Safety Guidelines and associated Sample Questions, issued during the year, and by the holistic safety self-assessment tool that is currently trialled by some licence holders. Pilot assessments will include ARPANSA's Radiation Health Services Branch and Medical Radiation Services Branch. Looking ahead, I believe the application of the holistic safety assessment guidance and tools will provide ARPANSA with a deeper understanding of the safety culture among licensees as well as foster an improved safety culture.
Other key Agency activities in this space include: periodic safety review of the OPAL Reactor; review of ANSTO's emergency preparedness and response; and creation of a joint ARPANSA–Australian Safeguards and Non-Proliferation Office (ASNO) working group on nuclear security. These will all contribute to provide further insight into the safety and security of the operations of Australia's only nuclear facility.
A final remark on this topic is that ARPANSA's mandate is restricted to Commonwealth entities. However, ARPANSA has some insight in radiation safety more broadly through other mechanisms, such as the Australian Radiation Incident Register and the Australian National Radiation Dose Register, the latter developed and operated by ARPANSA to hold dose records for uranium mining workers. Reporting to the Incident Register is the responsibility of each jurisdiction, whereas the uranium industry is obligated to report occupational doses to the Dose Register as part of licence conditions. Data available to ARPANSA at this point in time do not indicate any negative trends.
SCIENCE
Level of scientific achievement – 10 significant projects completed, more than 20 papers published
Scientific and regulatory staff at ARPANSA participate in a large number of international and national scientific fora and projects. The output from these activities includes the production of standards, recommendations, guides, methodologies, evaluation and original scientific publications, all of which are listed in Appendix 9.
ARPANSA is a World Health Organization (WHO) Collaborating Center in Radiation Protection and the Agency participates in both the Intersun Project (on ultraviolet radiation health) and the Electromagnetic Radiation Programme. ARPANSA, along with the Peter MacCallum Cancer Centre, participates in the Radiation Emergency Medical Preparation and Assistance Network. All of these activities provide ARPANSA with an excellent opportunity to interact with the scientific and health protection communities in areas of core relevance to the Agency's programs. ARPANSA staff also regularly participate in Task Groups that develop the recommendations of the International Commission on Radiological Protection (ICRP). From 1 July 2013, I will be a member of ICRP's Main Commission as well as Chair of Committee 5 (on environmental protection). Over the year, ARPANSA has participated in developing recommendations relating to radon progeny, geological disposal of radioactive waste, and protection of the environment under planned, emergency and existing exposure situations.
The United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) reports annually to the United Nations General Assembly on sources and effects of ionising radiation. Australia has been a member of UNSCEAR since its inception in 1955. UNSCEAR is composed of representatives from twenty-seven United Nations Member States with support from alternate representatives and advisers. I am currently the Australian representative and Chair of UNSCEAR. At its 60th session, UNSCEAR agreed on the scientific conclusions in its scientific annex on Levels and effects of radiation exposure due to the nuclear accident after the 2011 Great East-Japan Earthquake and Tsunami, as well as in the annex on Effects of radiation exposures of children. Both studies will be published as scientific annexes to the 2013 Report to the United Nations General Assembly. ARPANSA also has the lead in UNSCEAR's ongoing collation of international data on exposure from medical uses of radiation.
SERVICE
Percentage customer satisfaction – greater than 80%
I note that in our most recent survey of licence holders, ARPANSA's performance as a regulator was rated excellent by 16%, good by 58%, and satisfactory by 26%. No licence holder rated our performance as either poor or very poor. Publication of our guidance documents rated particularly well as did our proportionate approach. Understandably, further reduction in application processing times would be appreciated.
Provision of services is a significant portion of ARPANSA's work. Services in terms of responding to telephone requests from members of the public, as well as regulatory services in the form of reviews and licensing, are not addressed here. However, the Agency provides specialised services in several areas, primarily the Personal Radiation Monitoring Service (PRMS) as well as the Australian Clinical Dosimetry Service (ACDS), calibration against the Australian Primary Standard for absorbed dose, and the Ultraviolet Protection Factor (UPF) fabric testing.
The PRMS is a commercial activity; however it is also strategic for a number of reasons. It makes Australia independent of international service providers whose service may become scarce in case of an emergency; it is capable of supporting ARPANSA staff and responders with personal radiation monitoring in case of a national emergency; and, it broadens the basis of expertise in dosimetry in the Agency – noting that dosimetry is a core competency in radiation protection. A major reinvestment program has commenced during the year, to be continued over several years, to modernise the service and adapt to customer needs.
The ACDS performs audits of linear accelerators (or linacs) used in therapy centres across Australia. The ACDS was established under a Memorandum of Understanding between ARPANSA and the Department of Health and Ageing which commenced in 2011 and the Memorandum of Understanding will come to an end in the financial year 2013–14. The service is well on track in relation to the targets defined in the Memorandum of Understanding. It has also delivered pre-clinical audits to several centres that were not foreseen in the Memorandum of Understanding and has been very well received by the radiotherapy community. Its activities are currently audited by KPMG and I look forward to discussions with the Department of Health in the next year regarding future activities in this area.
ARPANSA holds the primary standard for absorbed dose in Australia; the secondary standards that are used for calibration in radiotherapy centres across Australia are traceable to the ARPANSA standard. In the future, ARPANSA will move towards direct calibration of secondary standards using its medical linac, and aim at gradually reducing the reliance on cobalt-60 calibration.
The UPF Testing Service evaluates the ultraviolet (UV) protection properties of fabric used for clothing. Garments with an approved UV protection rating may be issued with a ‘swing tag' with the relevant UV protection factor indicated. Approximately five million swing tags are issued every year. The service is unique for Australia. The UPF testing area carries out client surveys bi-annually, the last one was in November 2012. No formal customer satisfaction survey has been performed this financial year.
The PRMS has grown slightly and the UPF Testing Service has been fairly stable. The calibration of secondary standards has increased slightly. The ACDS has been met with overwhelmingly positive responses. I conclude that the service provision has been met by a positive response in general; ongoing upgrading as well as improved customer surveys should lead to enhanced understanding of customer needs and improved service delivery.
FINANCE
Operation within budget
ARPANSA operated within its available financial resources during 2012–13. We report an operating deficit of $2.29m. This deficit is caused by depreciation and amortisation amounting to $2.33m for which no appropriation is needed. In total, we report a small surplus of $40k.
ARPANSA is committed to strong governance and the prudent management of resources in delivering its mandate to Government. The Agency continues to review all aspects of its delivery models to find ways of offering its services in a more efficient and economical manner. A number of innovative technological solutions were delivered during the financial year, consistent with this approach.
STAFF
Percentage staff satisfaction: more than 70%
Historically, ARPANSA has been characterised by very low staff turnover which, while encouraging stability, can potentially make reorientation of the Agency more difficult to achieve. In addition, total staff numbers have been decreasing in response to financial trends and to meet mandated-efficiency dividends. At end of this financial year, ARPANSA had 140.77 full time equivalents comprising a total number of employees of 149; of which 138 were ongoing and 11 were non ongoing.
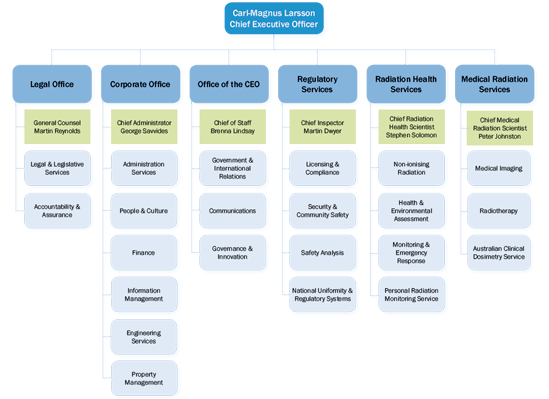
In order to sustain ARPANSA's delivery of health outcomes to the Australian Government and community, the Agency commenced a realignment towards the end of this financial year, which is expected to continue in the first part of the 2013-14 financial year. The term ‘realignment' was chosen to differentiate it from an earlier major Agency restructure occurring in early 2011, noting that the realignment was designed to meet requirements of the Strategic Directions 2012-16. Whilst not as dynamic as the 2011 restructure, this realignment still affected about a quarter of the Agency's staff. The resulting organisational structure as outlined in Figure 1 took effect from 30 June.
Figure 1: Organiation chart
(click on image for larger view)
ARPANSA took part in the 2013 APS employee census. Participation was extraordinary, 86 % compared to the APS average of 66%. As a matter of fact, ARPANSA ranked number four of all 113 APS agencies captured in the census. Whilst the high participation rate in itself does not tell us anything about the level of satisfaction, I still consider it an indicator of a very healthy engagement.
A fulsome analysis of the census data was not available at the end of June and analysis is still ongoing at the time of preparation of this report. However, the APSC has provided us with some preliminary data. It is satisfactory to see that some of the descriptors related to general satisfaction rate very highly, in many cases above 80% and often above the APS average and above the previous years. Some descriptors rank less well, including performance management and management in general – however, also in these areas there are clear signs of improvement which is likely the result of a substantial investment over the year in executive leadership development including performance management that has been made available for staff from APS6 to Senior Executive Service level, and commencement of education for all staff with a focus on expectations of the Public Service.
Outlook for 2013–14
I intend to take every opportunity to work closely with my staff to continue to deliver a professional service to the Australian Government and community. Significant activities planned for the coming year include, but are not limited to, the following:
- Improving regulatory practice including further consideration of observations from reviews and audits
- Working with the Australian National Audit Office (ANAO) in their performance audit of the Agency's regulatory activities and taking action on resulting recommendations
- Continuing the implementation of ARPANSA's Incident Management Plan and establishing links to the Business Continuity Plan and other important risk mitigation strategies, with a view to execution of the plans in the future
- Working with the Portfolio Department on the sustained delivery of high-quality and well-audited therapy services across Australia
- Expanding work on diagnostic reference levels to new modalities which provide a tool for radiological facilities to assist in their cycle of quality improvement
- Reaching final decisions in relation to applications for a radiopharmaceuticals production facility, a waste treatment facility, and an interim store for intermediate level waste, submitted by ANSTO
- Finalising ARPANSA's assessment of the Periodic Safety Review of OPAL and the Emergency Preparedness and Response Review of ANSTO
- Enhancing the development of codes and guidance within the national uniformity process
- Continuing the assessment of scientific literature and guidance on exposure to extremely low frequency electromagnetic radiation, magnetic fields and radiofrequency electromagnetic radiation, revitalising stakeholder consultation in this area and revising standards as necessary
- Continuing promotion of international safety and security in line with Australian obligations and priorities which includes reporting under the Convention on Nuclear Safety and under Code of Conduct on the Safety and Security of Radioactive Sources
- Continuing and expanding the interactions with the scientific community, nationally and internationally
- Further developing procedures for planning and internal accountability, including commencing work towards a certification of Agency operations according to ISO 9001
- Continuing the work towards a high-performing Agency with motivated and competent staff working in an environment that is conducive of both performance and satisfaction.
My assessment is that ARPANSA has a unique blend of staff with very high competence in many of the areas that are core to the Agency's delivery. We are, however, experiencing shortage in some areas and looking ahead, I can see that it will be difficult to replace some long-service experts when they retire, as the basis for this specialised knowledge in Australia, and elsewhere is thinning in important areas. I am therefore looking into continued participation in the international regulatory and scientific frameworks, establishing closer collaboration with expertise both nationally and internationally, and to recruit strategically as senior experts retire.
In conclusion, ARPANSA is an agency with a highly committed and very capable and competent workforce committed to continuous improvement; it is in a good position to tackle the challenges ahead.
1 The High Flux Australian Reactor (HIFAR) operated between 1958 and 2007, when it was permanently shut down.


