Licence application
In October 2012, the Australian Nuclear Science and Technology Organisation (ANSTO) announced its intention to expand its Molybdenum-99 (Mo-99) production with the construction of a new Mo-99 facility at Lucas Heights. Molybdenum-99 is an important radioisotope used in nuclear medicine for the diagnosis of a wide variety of medical disorders and ANSTO is a major national and international supplier.
In its ANM Mo99 site licence application submitted to ARPANSA on 25 October 2012, ANSTO announced its intention to treat and manage the intermediate level liquid waste to be generated from the ANM Mo99 through an immobilisation process applying SYNROC technology at the ANSTO SyMo Facility. SYNROC (synthetic-rock) is an ANSTO-developed technology to transform the liquid waste generated from the Mo-99 production process into a stable immobilised glass ceramic form. The technology greatly reduces the waste volume and converts it into a long-term stable form. The application for the ANSTO SyMo facility was received on 6 August 2012 and a construction licence was granted by ARPANSA on 13 May 2014. Proposals for both facilities were referred to the Department of Sustainability, Environment, Water, Population and Communities for assessment under the Environmental Protection and Biodiversity Conservation Act 1999 (EPBC Act). The Department of Sustainability, Environment, Water, Population and Communities determined that no environmental assessments are required for these facilities under the EBPC Act:
(ANSTO)/Commonwealth/Lucas Heights/NSW/Synroc Waste Treatment Facility
ANSTO/Science and research/Lucas Heights/NSW/ANSTO Nuclear Medicine Mo99 Facility, NSW
Under the Act, ARPANSA’s CEO is required to assess the applications and decide whether or not licences should be granted.
Public consultation
In line with the Act, the CEO of ARPANSA placed a notice of intention to make a decision on the ANSTO Nuclear Medicine Molybdenum-99 facility siting licence application on the ARPANSA website and advertised this via national and local daily newspapers, and the Public Service Gazette.
In this notice ARPANSA also provided an invitation to the public to make submissions on the licence applications. Submissions closed on 12 June 2013.
ARPANSA also held a community information session at the Engadine Community Centre on 16 May 2013.
The ANSTO SyMo facility is a prescribed radiation facility under regulation 6 of the Australian Radiation Protection and Nuclear Safety Regulations 1999. Although it is not mandated in the Regulations to invite submissions from the public during the licensing of such facility, the application was included in the consultation process established for the ANSTO Nuclear Medicine Molybdenum-99 facility.
The Community Information Session also covered applications for the siting and construction of the ANSTO interim waste store.
Details of the applications and submissions received are available on our Public consultation webpage.
Decision on licence application
On 4 October 2013, the CEO of ARPANSA decided to issue a licence to the Australian Nuclear Science and Technology Organisation (ANSTO) to prepare a site for a controlled facility (the ANSTO Nuclear Medicine Molybdenum-99 Facility) at the ANSTO Lucas Heights Science and Technology Centre. The CEO's Statement of Reasons and the Regulatory Assessment Report are available via the following link:
CEO decision - ANSTO nuclear medicine molybdenum-99 facility
On 13 May 2014, a delegate of the CEO of ARPANSA decided to issue a licence to the Australian Nuclear Science and Technology Organisation (ANSTO) to site and construct a prescribed radiation facility (the ANSTO SyMo Facility) at the ANSTO Lucas Heights Science and Technology Centre. The CEO’s Statement of Reasons and the Regulatory Assessment Report are available via the following link:
Delegate of the CEO decision – ANSTO SyMo facility
Application and review process
ARPANSA assesses applications based on the process shown in the following diagram:
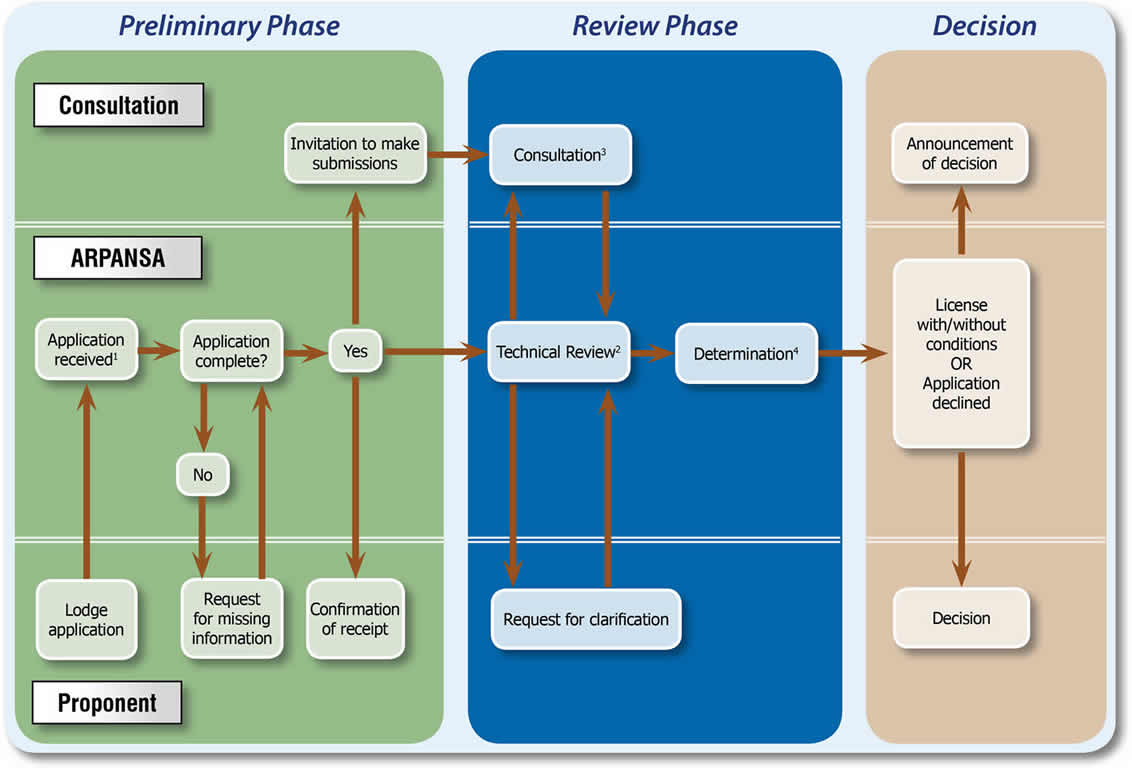
Notes:
1Application
The licence application may be for an individual phase or may cover more than one phase. Licences are required for each of the phases:
(a) Siting
(b) Construction
(c) Operation
Note that the ANM Mo99 facility licence application is for siting only. For more information on the ANM Mo99 construction licence applicaton see ANSTO Mo99 and SyMo application information. The ANSTO SyMo facility licence application is both to site and construct a prescribed radiation facility.
ARPANSA has issued application guides for a prescribed radiation facility and a nuclear installation which can be found at Regulatory guides.
2Technical review
The issues to be considered during the technical review include, but are not limited to, the following:
(a) Health and Safety of people and the environment, including whether doses are as low as reasonably achievable
(b) Net benefit
(c) Public submissions
(d) Requirements under the Australian Radiation Protection and Nuclear Safety Act and Regulations
(e) International Best Practice
(f) Contingency arrangements
(g) Plans and arrangements for operation of the facility which detail:
- Effective control
- Safety management plan
- Radiation protection plan
- Radioactive waste management plan (for waste that might arise during operations)
- Ultimate disposal or transfer
- Security plan
- Emergency plan
(h) Preliminary safety analysis report
(i) Operating limits and conditions.
3Consultation
The Regulations require ARPANSA to undertake a community consultation process for nuclear installation licence applications. While the ANSTO SyMo licence application related to a prescribed radiation facility, such a facility is not the same as a nuclear installation for the purposes of the Act and Regulations, however, the CEO decided to include the ANSTO SyMo licence application in the community consultation process being conducted for the ANM Mo99 facility licence which did relate to a nuclear installation. The consultation process involves the following steps:
(a) Once an application is received, and considered complete for the purpose of commencing its review (including public consultation), the CEO of ARPANSA must publish a notice advising of the intention to make a decision on the application in:
- the nationally circulated daily newspapers The Financial Review or The Australian
- the Government Gazette
The CEO may also publish notices advising of his intention to make a decision on the application in:
- a Sydney metropolitan daily newspaper
- a local newspaper.
(b) The CEO will also use this notice to invite members of the public and other bodies to make submissions about the application (within a fixed timeframe and provide procedures for making submissions).
(c) When seeking public submissions about the application, the CEO will also make the application public on the ARPANSA website and be open to requests for further consultation processes (such as public hearings).
(d) Public hearings, panels or other forms of community engagement may be organised after the submission time and be based on some of the concerns raised in the submissions. ARPANSA may invite additional experts to provide information as required.
(e) ARPANSA will accept submissions in any form. While electronic submission (via website or email) is preferable, ARPANSA will accept other forms including written and verbal.
(f) The proposed public submission period is one month.
4Determination
The decision to grant a licence, to grant a licence with conditions, or to decline the application will take a minimum of three months. The exact time is dependent on the quality of the application and issues raised during the review.
Appeals of ARPANSA licensing decisions:
(a) Any decision by the CEO to refuse to grant a licence is reviewable under section 40 the Australian Radiation Protection and Nuclear Safety Act 1998.
(b) Stakeholders may also have rights under administrative law to seek review. See for example the Administrative Decisions (Judicial Review Act) 1977.
More information
To contact ARPANSA directly, see Contact us.


