Diagnostic reference levels
A diagnostic reference level (DRL) is an indicative measure used to assess whether, in routine conditions, the amount of radiation used is unusually high (or low) for a specified procedurei.
A DRL is not a regulatory limit, it is a benchmark that when exceeded triggers a review. Conducting a local dose audit and comparing the results to a DRL provides an imaging facility with a simple method of identifying situations where they are delivering an unusually high patient dose.
Modalities currently covered by Australian DRLs
ARPANSA has focussed on developing DRLs for the procedures with the highest dose burden on the Australian population. So far, DRLs have been published for:
- multi-detector computed tomography for adult and paediatric patients
- general nuclear medicine and PET for adult patients only
- CT conducted as part of SPECT/CT and PET/CT procedures for adult patients only
- diagnostic coronary angiography for adult patients only.
ARPANSA is also actively collecting data on other image guided interventional procedures (IGIP). There are no Australian DRLs for planar radiography, dental X-ray, cone beam CT or mammography.
The purpose of a DRL and why it's important
The main objective of a DRL is to help avoid excess radiation dose to patients for a specified imaging taskii.
A diagnostic reference level can be used to:
- promote an optimum range of doses for specified medical imaging protocols
- give you a common dose metric for the comparison of doses between facilities, for specified protocols and modalities
- prompt you to perform local dose audits (via regulatory requirements).
DRLs can only be effective if appropriate local review and action is undertaken when the doses observed are consistently outside the relevant diagnostic reference level. Although they can also provide some reassurance that current practice is consistent with industry norms.
How we determine DRLs
The International Commission on Radiological Protection (ICRP) recommends that DRLs should reflect common practice within a given geographical region. ARPANSA achieves this by determining DRLs based on the results of wide-scale national surveys of imaging facilities.
Survey participants submit their protocol, patient, and dose information to us for a variety of procedures. We use this information to calculate the facility reference levels (FRLs) for those surveys. The DRLs are based on the 75th percentile (third quartile) of the resulting FRL distributions.
More information for particular imaging modalities can be found in the relevant modality pages.
Facility reference levels
A facility reference level (FRL) indicates the typical patient dose and is the quantity you compare against the DRL. It is the median dose delivered to a sample of patients undergoing a particular routine diagnostic imaging protocol at a given facility. In most cases the dose is dependent on the equipment used to perform the imaging (for example in CT), and the FRL is also equipment specific (i.e. a facility may have more than one FRL for a single procedure).
FRLs can be used to:
- monitor your local facility doses for common procedures
- compare doses between similar protocols
- assess the dose impact of the introduction of new protocols
- compare doses between facilities
- compare with regional or national DRLs.
The following diagram shows the relationship between an FRL for a particular scan and the national DRL for that scan (in this case for a CT scan).
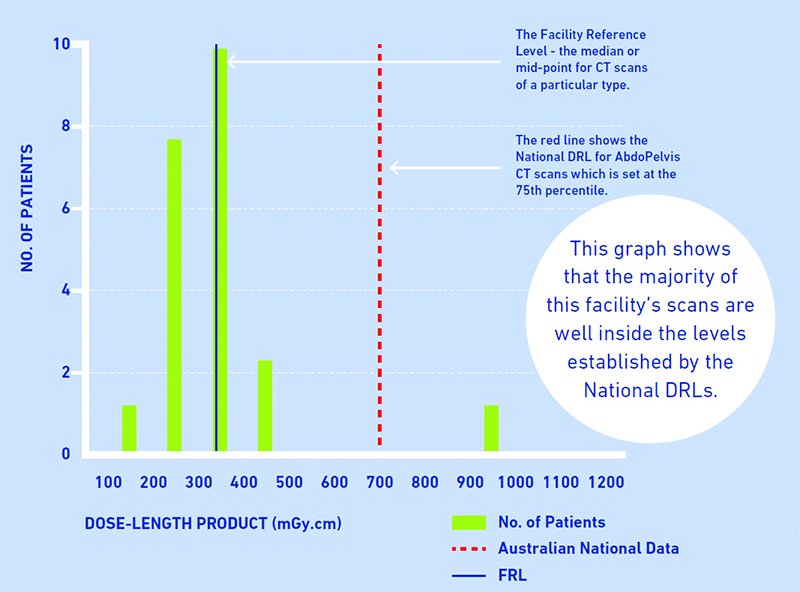
How you can calculate your FRLs
You should determine the median dose delivered to a representative sample of patients undergoing your common protocols. ARPANSA gives you a variety of tools to help with the calculation. To access these tools visit the relevant imaging modality pages.
If your FRLs exceed the DRLs
If your FRL exceeds the DRL for a particular protocol, it is an indication that you are delivering a higher dose than 75% of Australian imaging facilities for that procedure.
Section 3.2.15 of RPS C-5 states that you should review your imaging protocols if typical doses or administered activities for a representative sample of patients exceed the relevant diagnostic reference level. In practice, this means that if your FRL exceeds the DRL, you’ll need to review your protocols and procedures. If the review shows that you need to optimise your scans, you should perform this in collaboration with a medical physicist. The Australasian College of Physical Scientists and Engineers in Medicine (ACPSEM) has a register of qualified medical physicists and their areas of expertise on its website. This list is not exclusive and other medical physicists may have suitable qualifications and experience to advise on optimisation – check with your regulator if you have queries.
What you need to do
Know your regulatory requirements
Comparisons with diagnostic reference levels are required by state and territory regulatory bodies in accordance with an ARPANSA Code of Practice: either RPS C-5 (still to be adopted by some jurisdictions) or RPS 14. The two codes are in accordance concerning DRLs - i.e. meeting the newer RPS C-5 requirements will meet the RPS 14 requirements and vice versa. Section 3.2.15 of RPS C-5 states that:
The Responsible Person must establish a program to ensure that:
- radiation doses administered to patients for diagnostic purposes are compared with diagnostic reference levels (DRLs) at least annually for those radiological procedures for which DRLs have been established in Australia
- a review is conducted to determine whether the optimisation of protection and safety for patients is adequate, or whether corrective action is required, if, for a given type of radiological procedure:
- typical doses or administered activities for a representative sample of patients exceed the relevant diagnostic reference level or;
- exposures do not provide useful diagnostic information or do not yield the expected medical benefit to patients.
The Diagnostic Imaging Accreditation Scheme (DIAS), the Royal Australian & New Zealand College of Radiologists (RANZCR) Medical Imaging Accreditation Program (MIAP) and the Australian College on Healthcare Standards (ACHS) EQuIP 6 Accreditation Standards all require compliance with state and territory regulation, which in turn requires compliance with the ARPANSA Code of Practice.
Know your Diagnostic Imaging Accreditation Scheme requirements
The requirements of the Diagnostic Imaging Accreditation Scheme (DIAS) were amended in 2016 to include the following:
Your imaging practice must establish a program to ensure that radiation doses administered to a patient for diagnostic purposes are:
- annually compared with diagnostic reference levels (DRLs) for diagnostic procedures for which DRLs have been established in Australia.
- if DRLs are consistently exceeded, reviewed to determine whether radiation protection has been optimised.
Read the DIAS user guides and the advice on DRL comparison on their website.
Compare your FRLs to the DRLs at least once a year
As mentioned earlier, it depends on your state or territory regulator and DIAS auditors as to:
- how often you need to compare your FRLs to the DRLs
- what form that comparison takes.
However, it is ARPANSA's opinion that you should conduct a comparison annually and whenever new equipment or exposure parameters are introduced or if you notice something unusual. The exception is for general nuclear medicine procedures – see the nuclear medicine page for details.
Monitor your image quality
It is vital that image quality remains adequate for the diagnostic task. You should monitor the quality of images to ensure this. For CT scans, the RANZCR has a CT Image Review Self Audit protocol.
Even if you scan infrequently, compare to the DRLs
It is beneficial to compare even the relatively few doses from rarely performed scans to their respective DRL. Just keep in mind that if you have a small sample size then the characteristics of patients within your cohort may be the reason for an unusually high or low dose.
NDRLS principal stakeholders
- Australian Government Department of Health
- Diagnostic Imaging Accreditation Scheme
- The Royal Australian and New Zealand College of Radiologists
- Australasian College of Physical Scientists & Engineers in Medicine
- Australian state and territory regulators
- Australian and New Zealand Society of Nuclear Medicine
- Australian Society of Medical Imaging and Radiation Therapy
- Australasian Association of Nuclear Medicine Specialists
Contact us
If you have any further questions please contact us. We are available by telephone on 1800 033 972 between 9am and 4pm weekdays, or by email at ndrld@arpansa.gov.au.
i ICRP, 2017. Diagnostic reference levels in medical imaging. ICRP Publication 135. Ann. ICRP 46(1):1-143
ii ICRP, 2007. The 2007 Recommendations of the International Commission on Radiological Protection. ICRP Publication 103. Ann. ICRP 37(2-4):1-332


