Beta particles have a mass which is half of one thousandth of the mass of a proton and carry a single negative charge.
What are beta particles?
Beta particles (β) are high energy, high speed electrons (β-) or positrons (β+) that are ejected from the nucleus by some radionuclides during a form of radioactive decay called beta-decay. Beta-decay normally occurs in nuclei that have too many neutrons to achieve stability.
What are the properties of beta particles?
Beta particles have a mass which is half of one thousandth of the mass of a proton and carry either a single negative (electron) or positive (positron) charge. As they have a small mass and can be released with high energy, they can reach relativistic speeds (close to the speed of light).
Their light mass means that they lose energy quickly through interaction with matter and have a haphazard path as they move through air or other materials.
Beta particles are much less ionising than alpha particles and generally do less damage for a given amount of energy deposition. They typically have ranges of tens of centimetres in air (energy dependent) and a few millimetres in materials.
What causes some radionuclides to emit beta particles?
Beta minus particle (β-) emission occurs when the ratio of neutrons to protons in the nucleus is too high. An excess neutron transforms into a proton and an electron. The proton stays in the nucleus and the electron is ejected energetically.
This process decreases the number of neutrons by one and increases the number of protons by one. Since the number of protons in the nucleus of an atom determines the element, the conversion of a neutron to a proton actually changes the radionuclide to a different element.
Often, gamma ray emission accompanies the emission of a beta particle. When the beta particle ejection doesn't rid the nucleus of the extra energy, the nucleus releases the remaining excess energy in the form of a gamma photon.
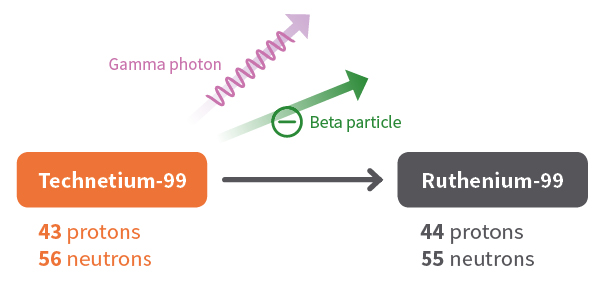
The decay of technetium-99, which has too many neutrons to be stable, is an example of beta decay. A neutron in the nucleus converts to a proton and a beta particle. The nucleus ejects the beta particle and some gamma radiation. The new atom retains the same mass number, but the number of protons increases to 44. The atom is now a ruthenium atom.
What are the health effects of exposure to beta particles?
Beta-particles, being less ionising than alpha-particles, can travel through many centimetres or even metres or air and through millimetres of skin or tissue. Sufficient intensity of beta-radiation can cause burns, rather like severe sunburn. If beta-emitting radionuclides are inhaled or ingested, they can also do damage to internal cells and organs.
What are some common sources of beta particles?
Many beta emitters occur naturally in the radioisotopes found in the natural radioactive decay chains of uranium, thorium and actinium. Examples include lead-210, bismuth-214 and thallium-206.
Beta emitters are also commonly found in the radioactive products of nuclear fission. Examples include strontium-90, caesium-137 and tritium.
What are some uses of beta particles?
The medium penetrating power of beta particles provides a range of useful applications which include:
- thickness detectors for the quality control of thin materials i.e. paper
- treatment of eye and bone cancers, strontium-90 or strontium-89 are commonly used
- Tritium is used in some phosphorescent lighting typically for emergency lighting as it requires no power
- Fluorine-18 is commonly used as a tracer for positron emission tomography (PET).


