A radioactive atom will attempt to reach stability by ejecting nucleons (protons or neutrons), as well as other particles, or by releasing energy in other forms.
Radioactivity is the property of some unstable atoms (radionuclides) to spontaneously emit nuclear radiation, usually alpha particles or beta particles often accompanied by gamma-rays. This radiation is emitted when the nucleus undergoes radioactive decay and is converted into a different isotope which may, according to its number of neutrons and protons, be either radioactive (unstable) or non-radioactive (stable). This “daughter” nucleus will usually be of a different chemical element to the original isotope
What causes atoms to be radioactive?
Atoms found in nature are either stable or unstable. An atom is stable if the forces among the particles that makeup the nucleus are balanced. An atom is unstable (radioactive) if these forces are unbalanced; if the nucleus has an excess of internal energy. Instability of an atom's nucleus may result from an excess of either neutrons or protons. A radioactive atom will attempt to reach stability by ejecting nucleons (protons or neutrons), as well as other particles, or by releasing energy in other forms.
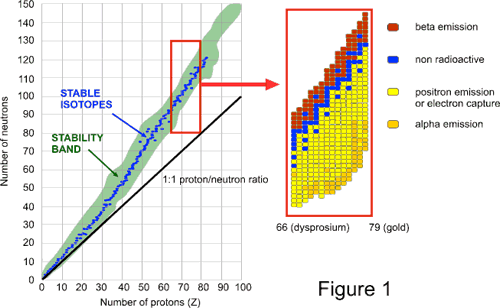
The band of nuclear stability (Figure 1) indicates various neutron/proton combinations that give rise to observable nuclei with measurable half-lives. A close-up look at the band of nuclear stability in the region from Z = 66 (dysprosium) through Z = 79 (gold) shows the types of radioactive processes undergone by various nuclides. Nuclides with lower neutron/proton ratios tend to undergo positron emission, electron capture, or alpha emission, whereas nuclides with higher neutron/proton ratios tend to undergo beta emission.
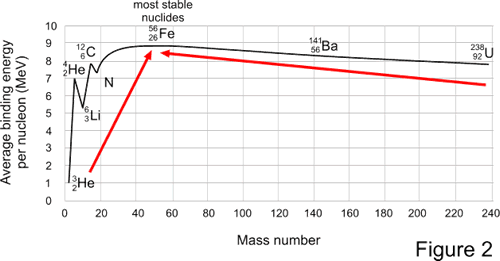
Another measure of stability is binding energy, the amount of energy needed to overcome the strong nuclear force and pull apart a nucleus. The binding energy per nucleon for the most stable isotope of each naturally occurring element is illustrated in Figure 2. Binding energy reaches a maximum of 8.79 MeV/nucleon at 56Fe. As a result, there is an increase in stability when much lighter elements fuse together to yield heavier elements up to 56Fe and when much heavier elements split apart to yield lighter elements down to 56Fe, as indicated by the arrows.
What happens to atoms after they release radiation?
As the nucleus emits radiation or disintegrates, the radioactive atom (radionuclide) transforms to a different nuclide. This process is called radioactive decay. It will continue until the forces in the nucleus are balanced. For example, as a radionuclide decays, it will become a different isotope of the same element if it gives off neutrons or a different element altogether if it gives off protons.
The series of transformations that a radionuclide goes through to reach stability and the type of radiation produced is characteristic of the radionuclide. The stages form a decay series.
What is the difference between radioactivity and radiation?
Radiation is the energy or particles that are released during radioactive decay. The radioactivity of a material refers to the rate at which it emits radiation.
The activity of a sample of radioactive material is determined by measuring the number of disintegrations per unit of time. A disintegration occurs each time a nucleus ejects particles or energy. Activity is measured in a unit called the becquerel - 1 becquerel is equivalent to 1 disintegration per second.
Is all nuclear radiation the same?
The radiation that emanates spontaneously from the nuclei of unstable isotopes (radionuclides) as the nuclei undergo radioactive decay is generally alpha, beta, or gamma radiation. Similar radiation may be produced artificially in particle accelerators or x-ray generators. The naming is complicated as the radiation is often named according to its source, even when identical to similar radiation coming from other sources. For example, high-energy electromagnetic radiation (photons) coming from the atomic electrons are called x-rays whereas similar photons coming from inside the nucleus are called gamma-rays.


